
This page is intended as a comprehensive and rapid survey of fatty acids possessing carbon–halogen covalent bonds, which have been described in living organisms, some of them being formed during chemical reactions in various industrial situations. Halogenated fatty acids which are one of the most interesting groups among the naturally occurring halogen compounds are not well known but several reviews on these compounds may be found. Halogenated fatty acids are found in different groups of organisms from microorganisms to the highest plants and animals. As halogen, they contain one or several atoms of fluor, chlorine, or bromine.
![]()
Fluorinated fatty acids as other lipids have been rarely identified in plants and microorganisms, while none has been identified in animals or marine organisms
The principal fluorinated component (3% of the seed oil) was detected in a west African shrub (Dichapetalum toxicarium, Chailletaceae) and identified as a ω-fluorooleic acid (18:1n-9) (Peters RA et al. Biochem Pharmacol 1959, 2, 25). D. toxicarium has also the ability to accumulate fluoroacetate in the leaves.
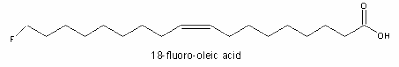
The toxicity observed in sheep eating the D. toxicarium seeds was explained by the metabolism of the fluoro acids which produces the very toxic fluoro-acetic acid.
Later, other fluorinated compounds were identified : ω-fluorocapric (C10:0), ω-fluoromyristic (C14:0), and ω-fluoropalmitic acids (Peters RA et al., Nature 1960, 187, 573). A reexamination of the seed oil after saponification and using mass spectrometry established in addition the presence of ω-fluoro derivatives of palmitoleic (C16:1), stearic (C18:0), linoleic (C18:2), and eicosenoic (C20:1) acids (Hamilton JTG et al., Chem Phys Lipids 2000, 105, 93). Details on these fluorinated fatty acids may be found in several reviews (Harper DB et al., Nat Prod Rep 1994, 11, 123; Dembitsky VM et al., Prog Lipid Res 2002, 41, 315).
An usual ω-fluoro-9,10-epoxy C18 fatty acid was identified together with other ω-fluoro acids of 16:1, 18:0, 18:1, 18:2, 20:0 and 20:1 in the seed oil of Dichapetalum toxicarium (Dichapetalaceae)(Hamilton JTG et al., Phytochemistry 1997, 44, 1129).
In plants, compositional profile of ω-fluoro acids containing triglycerides was shown to be similar to that of non-fluorinated triglycerides in terms of chain lenght and degree of unsaturation of esterfied fatty acids, suggesting that fatty acid synthetase is equally able to utilize fluoroacetyl-CoA and acetyl-CoA in the early steps of fatty acid synthesis.
Various halogenated fatty acids were chemically synthesized (review by Dembitsky VM et al., 2002).
A series of fluorine-containing unsaturated fatty acids, which are potential fungicides, have been prepared (Michel D et al., Synthesis 1996, 1007).
![]()
These compounds have been found to be the major constituents among organohalogens in fish, molluscs, invertebrates and seaweeds.
The presence of six isomeric acids, 9-Cl-9-, 10-Cl-9-, 9-Cl10-, 10-Cl-9, 11-Cl-12-, and 12-Cl-11-hydroxystearic acids were reported for the first time in jellyfish (Auritia aurita) (White RH et al., Biochemistry 1977, 16, 4944).
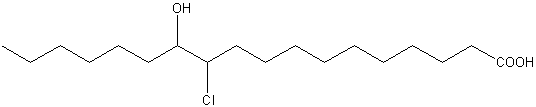
11-Cl,12-OH-stearic acid from Auritia aurita
All compounds, as the previous one, with adjacent chloro- and hydroxy- goups are named fatty acid chlorohydrins.
Later, several dichloro-tridecanoic, -hexadecanoic, and -octadecanoic acids were isolated from fish, mussel, lobster, or jellyfish.
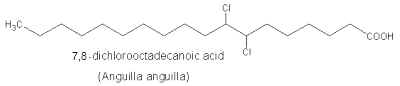
The occurrence of a tetrachloro-fatty acid was reported in fish lipids.
The formation of fatty acid chlorohydrins by the addition of HOCl to unsaturated fatty acids is well known (Winterbourn CC et al., Arch Biochim Biophys 1992, 296, 547). The reaction of HOCl in vitro with a variety of fatty acids and phospholipids (mainly plasmalogens) has been well studied (Wang WY et al., Anal Biochem 2013, 443, 148). Oleic acid yields approximately equal amounts of the 2 positional isomers of the 9,10-monochlorohydrin, while linoleic acid can form both monochlorohydrins and at higher HOCl/lipid ratios, bis-chlorohydrins (4 isomers are possible). With polyunsaturated fatty acyl chains, such as arachidonic, eicosapentaenoic and docosahexaenoic acids, increasingly complex mixtures of chlorohydrins can be formed. In phospholipids, such as glycerophosphocholines, multi-chlorinated products are formed, and increasing modification results in a greater tendency to generate lysophospholipids by hydrolytic cleavage of the modified fatty acyl chain (Arnhold J et al., Biochim Biophys Acta 2002, 1572, 91). These reactions suggest that the formation of lysophospholipids from unsaturated phospholipids by hypochlorous acid can be relevant in vivo under acute inflammatory conditions.
Thus, chlorinated lipids have been shown to be associated with multiple inflammatory diseases, including those affecting endothelial cells (Hartman CL et al., J Lipid Res 2018, 59, 113). It is known that 2-chlorofatty acids are produced as a result of myeloperoxidase-derived HOCl targeting plasmalogen phospholipids, thus promoting endothelial cell dysfunction, which may lead to broad implications in inflammation, thrombosis, and blood vessel stability. It has been also shown that 2-chlorofatty acids as a myeloperoxidase product are triggering the formation of neutrophil extracellular traps, which have been implicated in microcirculatory plugging following neutrophil activation (Palladino END et al., J Lipid Res 2018, 59, 1424-1432).
Chlorinated fatty acids are also formed during the production of bleached paper for oxidation of lignins using elemental chlorine. As chlorinated oleic and linoleic acids are found in the wood, several dichloro- and tetrachloro-acids are found in bleached pulp mill effluents. Similar compounds may be formed also during bleaching of wheat flour and during the disinfection process of drinking water.
A review of these fatty acids and their derivatives was made by Dembitsky VM (Prog Lipid Res 2002, 41, 315).
![]()
The first investigation of bromine in fatty acids was reported in the 1970s in lipid extracts from marine animals (Lunge G, JAOCS 1972, 49, 44).
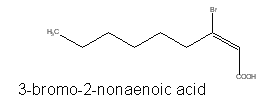
Several brominated fatty acids, such as 3-bromo-2-nonaenoic acid, have been isolated from red marine algae (McConnell O et al., Phytochemistry 1980, 19, 233).
Brominated fatty acids are rare in higher plants. Two brominated stearic acids, such as 9,10-dibromo-octadecanoic and 9,10,12,13-tetrabromo-octadecanoic acids, were isolated from the seed oil of the Asian plant Eremostachys molucelloides (Dembitsky VM, Prog Lipid Res 2002, 41, 315).
Lichens are unique symbiotic organisms that produce different brominated acetylenic acids such as 18-bromo-octadeca-5,17-diene-15-ynoic acid (Rezenka T et al., Phytochemistry 1999, 50, 97). Several other bromoallenic fatty acids have been found in lichen species. A short review on unusual brominated fatty acids may be found by Dembitsky VM (Inform 2003, 14, 30).
A bromoallenic acid has been characterized in lipids from lichens and may be used as chemotaxonomic purposes (Rezanka T. et al., Phytochemistry 2001, 56, 869). This triene compound is hydroxylated in 15 and brominated in 18.
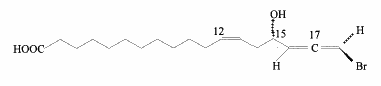
Similar molecules were described in sponges living in different locations and were named “demospongic acids”. They have been shown to have good antimicrobial activity (Hirsh S et al., Tetrahedron 1987, 14, 3257).
A series of brominated polyunsaturated long-chain fatty acids (C20-C28) has been isolated from different species of marine invertebrates (anemone, sponge) (Review in Dembitsky VM, Prog Lipid Res 2002, 41, 315).
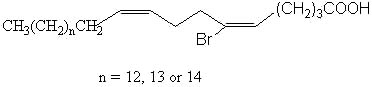
These unusual fatty acids were found in tropical marine sponges (Amphimedon terpenensis) and their structure elucidated by efficient techniques (Garson MJ et al., Lipids 1993, 28, 1011). The three fatty acids studied have a common scheme with the following structure:
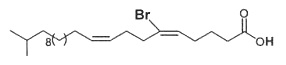
Fractionation of lipid extracts of a marine sponge Homaxinella sp led to the isolation of a new brominated fatty acid which was found in the free form or acylating a glycerol molecule. Physical studies revealed it has the structure of a branched-chain demospongic acid. The compound showed only moderate cytotoxicity (Mansoor TA et al., Lipids 2005, 40, 981).
DISPERSIVE LIQUID-LIQUID MICROEXTRACTION
Lire la suiteDevenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire