
PHOSPHOSPHINGOLIPIDS
WITH GLYCOSYL-CONTAINING
HEAD GROUPS
Complex phosphorylated glycolipids were discovered in 1958 by the group of Carter HE in soybean extracts (Carter HE et al., J Biol Chem 1958, 233, 1309) and during the 1960s in several plants (Carter HE et al., Biochemistry 1969, 8, 383). These compounds were named in 1958 "phytoglycolipids" and were shown to consist of ceramides (based mainly on phytosphingosine) linked to a phosphoinositol group which may be itself linked to a complex oligosaccharide. This later residue may be composed of inositol, mannose, fucose, arabinose, hexuronic acid, glucosamine, and xylose. The complexity and the diversity of these molecules led specialists to consider them as glycolipids more than phospholipids.
Further investigations have shown that these compounds are not restricted to plants but are found also in bacteria, yeast, fungi, and protozoans, while being absent in animals (Lester RL et al., Adv Lipid Res 1993, 26, 253). Thus, the trivial term "phytoglycolipids", used only in cosmetics, has been replaced by the generally accepted term inositol phosphorylceramides.
In bacteria (Sphingobacterium), a phosphomannose group was described instead of the phosphoinositol group present in the majority of the above cited organisms (Naka T et al., Biochim Biophys Acta 2003, 1635, 83).
Molecular species in each class differ in the structure and composition of the glycoside fraction but also in the composition of long-chain bases and fatty acids in the hydrophobic fraction. The most abundant long-chain bases are C18 and C20 phytosphingosine and dehydrophytosphingosine, and the most abundant fatty acids are hydroxy and non-hydroxy C24 and C26. In bacteria, the ceramide was shown to be composed of a branched C17 dihydrosphingosine and a branched C15 fatty acid (Naka T et al., Biochim Biophys Acta 2003, 1635, 83).
These phosphosphingolipids may contain :
1 – one glycosyl unit
2 – one oligosaccharide group with 2 or more sugar units
3 – one oligosaccharide group with a methyl-aminoethylphosphonic group
1 – Phosphosphingolipids with one glycosyl unit
These sphingolipids are mainly composed of inositol phosphorylceramide. They were reported to be a novel and major group of yeast sphingolipids since they form about one-third of the non-deacylable phospholipids in Saccharomyces cerivisiae (Smith SW et al., J Biol Chem 1974, 249, 3395). The ceramide moiety of these lipids, linked to a single phosphoinositol group, consists of phytosphingosine and a hydroxy C24 or C26 fatty acid.
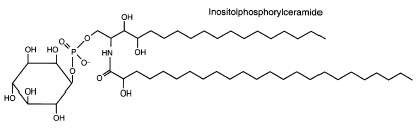
This structure is drawn with c18 phytosphingosine and hydroxy 24:0
Later, glycosylated phosphosphingolipids have been found in several fungi such as Phytophthora (Bruneteau M et al., Lipids 1997, 32, 359), Candida albicans (Wells GB et al., J Bacteriol 1996, 178, 6223) and Cryptococcus (Vincent VL et al., Microbiology 1995, 141, 1829). They were also found in various bacteria such as Flectobacillus (Batrakov SG et al., Biochim Biophys Acta 2000, 1484, 225) and Sphingobacterium (Naka T et al., Biochim Biophys Acta 2003, 1635, 83) and in yeasts (Dickson RC et al., Biochim Biophys Acta 1999, 1426, 347). Curiously, the inositol sphingophospholipid isolated from Sphingobacterium differs in its ceramide moiety from the other analogues found in the other abovementioned microorganisms since its long-chain base has a branched carbon chain (iso 17:0) as well as its fatty acid (iso 15:0).
A unique sphingophospholipid identified as ceramide phosphorylmannose was described in several species of Sphingobacterium, a bacteria known by the presence of high concentrations of sphingolipids as cellular lipid components (Naka T et al., Biochim Biophys Acta 2003, 1635, 83).
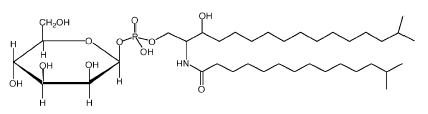
It was shown that these compounds activate murine macrophages via Toll-like receptor 4 (TLR-4) and stimulate bacterial clearance (Fujiwara N et al., Biochim Biophys Acta 2013, 1831, 1177). Interestingly, this TLR 4-recognition pathway of bacterial sphingophospholipid involves the fatty acid composition of ceramide in addition to the sugar moiety. Thus, a non-hydroxy fatty acid composed of ceramide was necessary to activate murine macrophages.
2 – Phosphosphingolipids with an oligosaccharide group
In these more complex sphingolipids, the oligosaccharide chains are commonly linked to C-2 and/or to C-6 of inositol and contain glucosamine acetylated or not, glucuronic acid, fucose, and different amounts of arabinose, galactose, and mannose. These compounds are frequently named phytoglycolipids.
Among a wide range of structures, the simplest forms are found in yeast and fungi, more complex forms are found in protozoa and in plants. While the involvement of these phosphorylated sphingolipids in the field of antifungal chemotherapy led to extensive studies in yeast and fungi, plants await further studies of their structures, metabolism, and functions.
Some examples of the diversity of these compounds found in yeast, fungi, plants and animals are given below (review in Lester RL et al., Adv Lipid Res 1993, 26, 253 and Dickson RC et al., Biochim Biophys Acta 1999, 1426, 347).
Abbreviations used : Cer : ceramide, Fuc: fucose, Gal: galactose, GlcNH2: glucosamine, Ins: inositol, Man: mannose, P: phosphodiester,
2.1 – YEAST AND FUNGI
Some examples among the numerous structures described in yeast, filamentous fungi, and mushrooms are shown below:
Cer – P – Ins – Man (1 to 5 residues)
in the yeast Saccharomyces (Smith SW et al., J Biol Chem 1974, 249, 3395), the fungi Sporothrix (Toledo MS et al., Biochem Biophys Res Comm 2001, 280, 19), Aspergillus (Bennion B et al., J Lipid Res 2003, 44, 2073) and in higher mushrooms (Jennemann R et al., Eur J Biochem 2001, 268, 1190).
Cer – P – Ins – Man – P – Man – oligomannoside (1 to 19 Man residues)
known as "phospholipomannan" in Candida (Trinel PA et al., J Biol Chem 2002, 277, 37260).
Cer – P – Ins – GlNH2 – Man – Man
in Sporothrix (Toledo MS et al., FEBS Lett 2001, 493, 50).
Cer – P – Ins – P – Ins
in the fungus Neurospora (Lester RL et al. J Biol Chem 1974, 249, 3388)
Cer – P – Ins – Man – P – Ins
in the fungus Candida (Wells GB et al., J Bacteriol 1996, 178, 6223) and in the yeast Saccharomyces (Lester RL et al., Adv Lipid Res 1993, 26, 253)
Cer – P – Ins – Man – Gal – Fuc – Gal (1 to 3 residues)
in a higher mushroom Agaricus (Jennemann R et al., Eur J Biochem 1999, 259, 331).
Cer – P – Ins – Man – Gal – Man
in the mycopathogen Paracoccidioides (Levery SB et al., Biochemistry 1998, 37, 8764).
2.2 – PROTOZOA
A core structure was described in several protozoa :
Cer – P – Ins – GlcNH2 – Man
This structure may be elongated with other mannose units and substituted with 2-aminoethylphosphonate (-O-P-O2CH2CH2NH3) on glucosamine as in Trypanosoma (Previato JO et al., J Biol Chem 1990, 265, 2518) or on glucosamine and mannose as in Leptomonas (Previato JO et al., J Biol Chem 1992, 267, 24279). These forms were named "lipopeptidophosphoglycan".
A sphingolipid having as a core structure :
Cer – P – Ins – Fuc
with the inositol moiety substituted with phosphoethanolamine was described in Trichomonas (Singh BN et al., Biochem Biophys Res Comm 1988, 157, 1239).
The evidence of complex structures was offered in other protozoa (Leishmania, Acanthomoeba) but they were only partially characterized (Lester RL et al., Adv Lipid Res 1993, 26, 253).
It is noteworthy that the core structure :
Cer – P – Ins – GlcNH2 – Man
found in several sphingolipids of yeast, fungi and protozoa and even in plants (Oxley D et al., PNAS 1999, 96, 14246), as shown above, is also found in the complex membrane structures known as glycosylphosphatidylinositol anchors.

For that reason, studies of the African trypanosome have played a great role in the development of the knowledge of the structure and function of membrane anchors in the 1980s (Ferguson MA, J Cell Sci 1999, 112, 2799).
2.3 – PLANTS
If studies on inositol phosphorylceramides have begun in the 1950s, it must be noticed that only few plants have been examined for the presence, structure, and function of these specific lipids.
Detailed structural analysis was first carried out on the sphingolipids isolated from corn seed (Carter HE et al., J Lipid Res 1969, 10, 363). The core structure was determined to be :
Cer – P – Ins – Man
but with the inositol moiety substituted with a glucuronic acid-glucosamine group.
Further studies on the tobacco leaves were carried out and led to the description of more additional components (Kaul K et al., Biochemistry 1978, 17, 3569 and Hsieh TC et al., J Biol Chem 1981, 256, 7747). Their core structure was determined to be :
Cer – P – Ins – Glucuronic acid – Acetylglucosamine
Cer – P – Ins – Glucuronic acid – Glucosamine
some of them are substituted with galactose or an arabinose-galactose group.
A hexose-hexuronic-inositol phosphoceramide was shown to be the major glycolipid species present in the leaves of Arabidopsis, while N-acetyl-hexosamine instead of hexose was shown to be present in tomato and soybean leaves (Markham JE et al., J Biol Chem 2006, 281, 22684).
2.4 – INVERTEBRATES
A novel series of phosphorylated glycosphingolipids with a choline phosphate group as an oligosaccharide substituent has been discovered in an Annelida earthworm, Pheretima hilgendorfi (Sugita M et al., J Biol Chem 1992, 267, 22595).
The structural elucidation of the oligosaccharide moieties enabled the description of two different structures as follows:
Cer – Gal – P – choline
Cer – Gal – Gal – P – choline
The ceramide moieties were mainly composed of either 22:0 or 24:0 linked to octadecasphingosines or nonadecasphingosine homologues.
3 – Phosphonoglycosphingolipids
These particular glycolipids contain the characteristic phosphonic C-P bond instead of the classical C-O-P bond. The phosphonic group is for the majority of compounds N-methyl-aminoethylphosphonic acid, aminoethylphosphonic group being present in very low amounts. Both groups are bound via an ester bond to the hydroxy group at the C-6 of galactose.
Several species with one phosphono group and various numbers of sugar moieties (with or without fucose) have been described in sea water molluscs (review in Hori T et al., Prog Lipid Res 1993, 32, 25).
Devenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire