
They form a large group of natural products which includes steroids and consequently sterols, they derived from C30 precursors.
Triterpenes, among numerous pharmacological properties, exhibit immunomodulatory function by various mechanisms. Different triterpenes, including components of commonly consumed plants, can promote some protection and alleviation of disease symptoms linked with immune responses and thus enhance overall well-being (review in: Renda G et al., Phytochemistry Reviews 2022, 21, 537). The triterpenes have been reported to possess a wide range of therapeutic applications including anti-viral properties which have been summarized in a review (Darshani P et al., Phytochem Rev 2022, 21, 1761–1842).
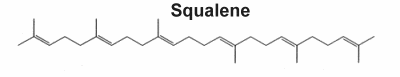
Nearly 200 different triterpene skeletons are known from natural sources and represent structurally cyclization products of Squalene which is the immediate biological precursor of all triterpenoids. In 1934, Robinson proposed a direct cyclization of squalene to the steroid molecule. In 1936, Nobel laureate researcher Paul Karrer described the biochemical structure of squalene for the first time. It has major specificities, which are related to anticancer properties, the maintenance of the oxidation/antioxidation balance, and its antiaging capabilities (review in Ronco AL et al., Funct Foods Health Dis 2013, 3, 462). Shark liver oil is considered the richest source of squalene, accounting for at least 40% of its weight. It is also widely distributed in nature, in lesser proportions in amaranth oil (6-9%), in wheat germ oil, and in olive oil (usually from 0.4% up to 1% in extra virgin olive oil). It has been shown that the human body synthesizes an average amount of 1.5 g/day (Liu GC et al., J Lipid Res 1976, 17:38).
Biochemistry, molecular biology, and applications of squalene (Spanova M et al., Eur J Lipid Sci Technol 2011, 113, 1299), and the origin of their skeletal diversity (Xu RX et al., Phytochemistry 2004, 65, 261) have been reviewed.
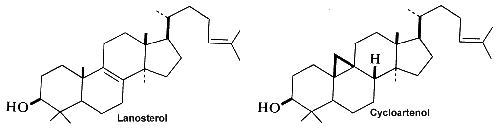
Squalene epoxide (2,3-oxidosqualene) is produced by the enzyme squalene epoxidase which use NADPH and oxygen to oxidize squalene. This metabolic step is the first in sterol biosynthesis leading to the formation of lanosterol or cycloartenol.
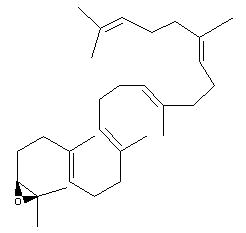
Squalene epoxide
Squalane is a completely saturated derivative of squalene. Present in sebum, it is largely used as a component in many cosmetic products. It is obtained by hydrogenation of squalene extracted from olive oil.
The large group of steroids, including sterols, are present in very small amounts in bacteria but at larger amounts in plants and animals while the hopanoids are very abundant in prokaryotes where they replace cholesterol.
Triterpenoids are widely distributed in edible and medicinal plants and are an integral part of the human diet. They are being evaluated for use in new functional foods, drugs, cosmetics and healthcare products. Screening plant material has identified fruit peel and especially fruit cuticular waxes as promising and highly available sources (Szakiel A et al., Phytochem Rev 2012, 11, 263). Amyrin isomers are the predominant compounds in tomato fruit wax, with quantities that vary considerably in different cultivars and during the subsequent stages of fruit development. A methyl ether derivative of an amyrin isomer, miliacin, has been proposed as a sedimentary molecular tracer of the history of Panicum (broomcorn millet) cultivation (Bossard N et al., Org Geochem 2013, 63, 48).
A review on plant triterpenoids with bond-missing skeletons, their biogenesis, distribution and bioactivity may be consulted (Almeida et al., Nat Prod Rep 2020, 37, 1207).
Protostane is a tetracyclic triterpene, primarily limited to the genus Alisma. It is so named because it is considered to be the “prototype” of steroids.
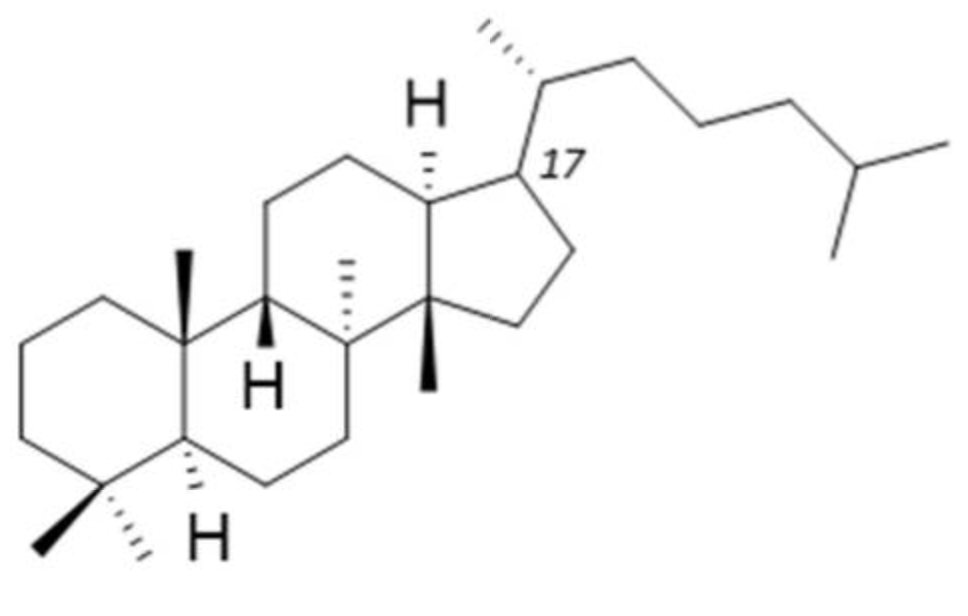 Protostane
Protostane
Alisols are the main subgroup of triterpenes found in Alismatis rhizoma. They form a family of 22 compounds designated alisol A to alisol V, the leading compoundsbeing alisol A and its epoxide derivative alisol B.
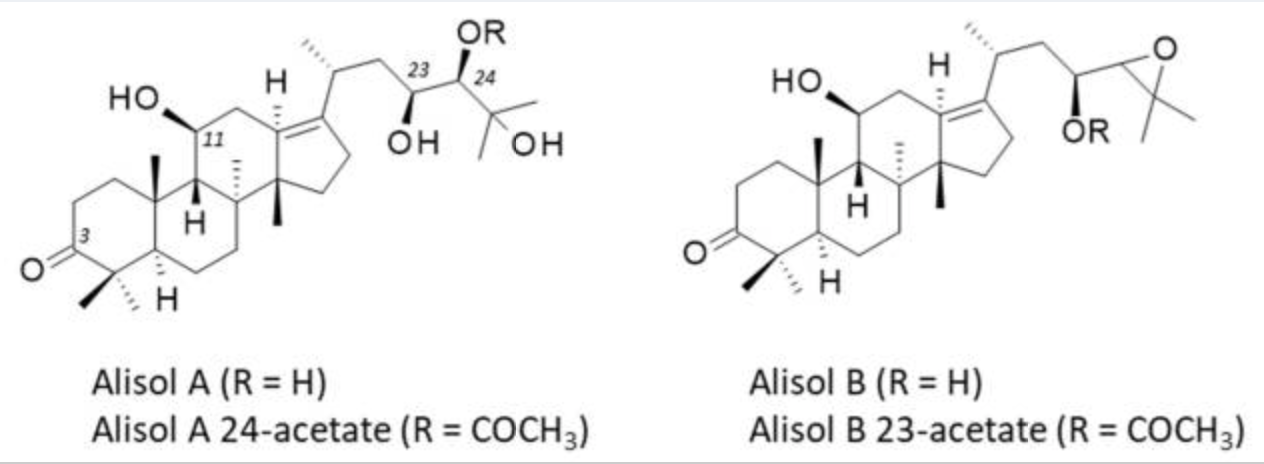
Both alisols display marked anti-inflammatory and anti-proliferative properties, useful for the treatment of diverse human pathologies, including metabolic disorders and cancers. In the present review, The main pharmacological properties of these bioactive natural products have been rewed (Bailly C et al., Biomedicines. 2022, 10(8), 1945).
Another important triterpenoid is asiatic acid. It is an active molecule from Centella asiatica (Apiaceae) used in dermatose treatment, it is one of the most active cicatrizing compounds. It was shown that asiatic acid can ameliorate intracellular oxidative stress caused by chemotherapy drugs (Welbat JU et al., Nutrients 2018, 10(8). pii: E1053). Some studies have indicated the potential of asiatic acid to induce autophagy in cancer cells through changes in the levels of specific proteins. It can also act as an anti-tumor immunotherapeutic agent (Wiciński M et al., Cancers (Basel) 2024, 16(7), 1317).
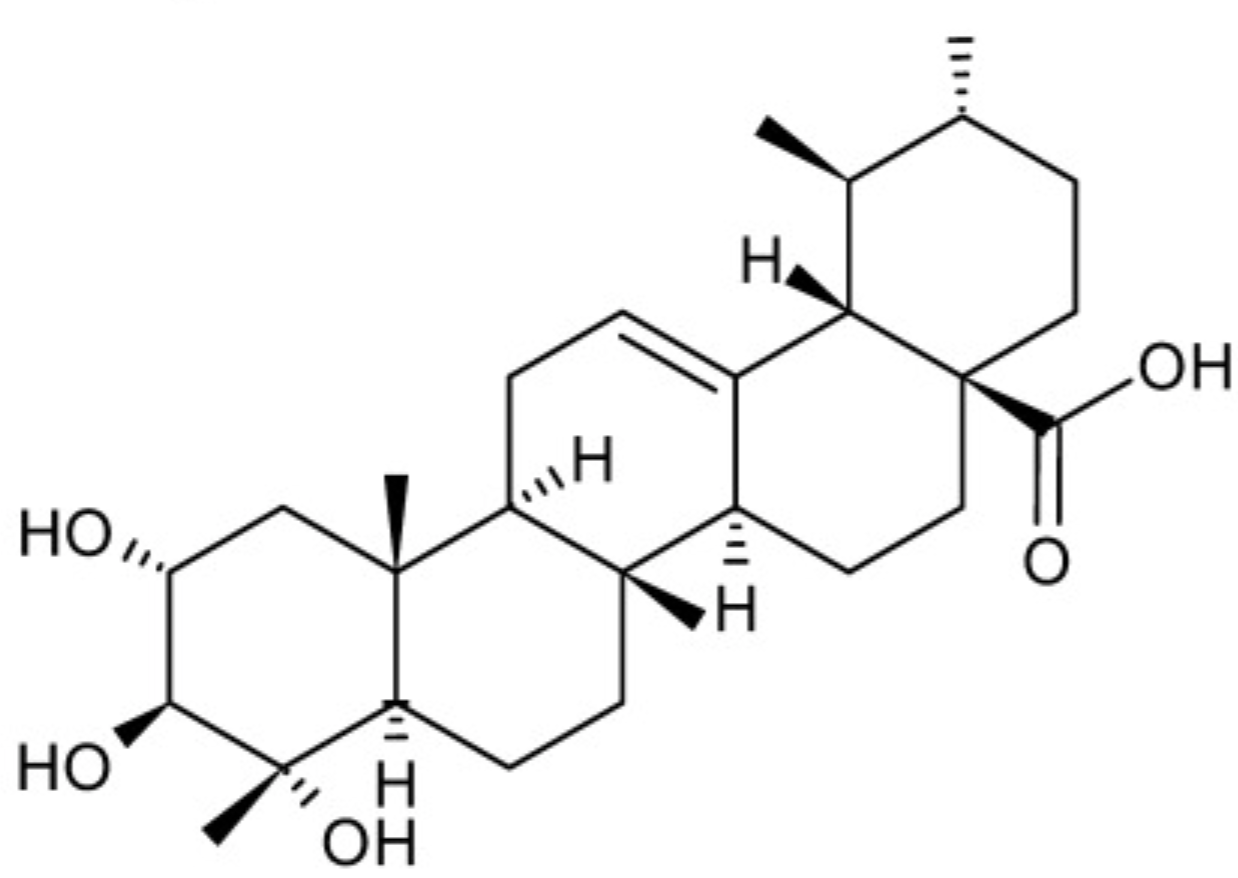 Asiatic acid
Asiatic acid
Pomolic acid (or benthamic acid) was first isolated from the peels of apples. The compound is commonly reported in species of the families Rosaceae and Lamiaceae. Anti-cancer activities represent its major pharmacological properties with breast cancer and leukemia cells being most susceptible. A wide array of other pharmacological properties of pomolic acid have
been reported (Chan EW et al., J Appl Pharmaceut Sci 2026, 13, 58). More recent findings have shown that it is an antagonist and selective modulator of peroxisome proliferator-activated receptor gamma (PPARγ). It reduced lipid accumulation during 3T3-L1 adipocyte differentiation and antagonized rosiglitazone-induced PPARγ transactivation without intrinsic agonist activity (Nicola-Llorente M et al., J Agric Food Chem 2026, 74/18, 14376).
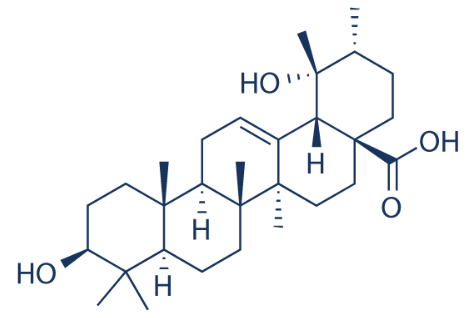 Pomolic acid
Pomolic acid
Medicagenic acid (or castagogenin) is a triterpenoid isolated from Medicago species but also from several plant products. It exhibits potent fungistatic effects against several plant pathogens and human dermatophytes. It has also enzyme inhibitory activities, the targets being xanthine oxidase, collagenase, elastase, tyrosinase. Interestingly, it showed the strongest predicted interaction with hyaluronidase with a high docking score (Noreen S et al., Food Sci Nutr 2026, 14, e71844), thus being a candidate for further investigation in dermatological and functional food applications.
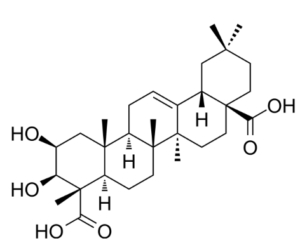 Medicagenic acid
Medicagenic acid
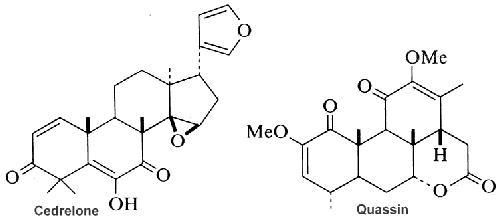
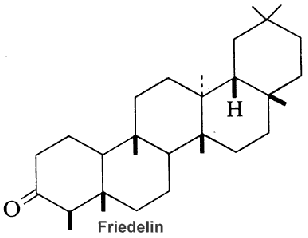
Among the large number of triterpenoid structures, some of them are shown below lanosterol, cycloartenol, cephalosporin, cyclobuxine, cedrelone, quassin, friedelin.
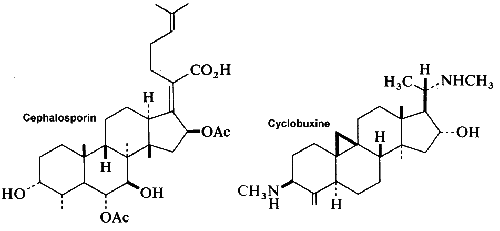
Argentatins are present in the resin of gayule (Parthenium argentatum), an Asteraceae cultivated for natural rubber production in the south of USA. They are tetracyclic-type triterpenoids synthesized from the C30 oxygenated cycloartane or lanostane skeleton (Xu Y et al., ACS Omega, 2021, 6, 15486), and are the main components of guayule resin, representing 20–30% of total biomass. A total of 12 new cycloartane- and lanostane-type triterpenoids were isolated from the resin of Parthenium argentatum AZ-2 obtained as a byproduct of Bridgestone guayule rubber production. The substantial abundance of argentatins in guayule leaves suggests the potential for future exploitation (Garcia Martinez MM et al., Plants 2023, 12, 2021). Argentatin A has antimicrobial activity against pathogenic bacteria, anti-inflammatory activity, and cytotoxic activity against a broad panel of human cancer cell lines.
 Argentatin A
Argentatin A
Cedrelone belongs to the group of limonoids (C-26 terpenoids related to limonin) which was isolated from a Brazilian native plant Trichilia catigua (Meliaceae), commonly used in popular treatments for several diseases. It induces cancer cell apoptosis, and possess antitumor effects.
Chemically, the limonoids consist of variations of the furanolactone core structure. The prototypical structure of limonoids consists of four six-membered rings and a furan ring. They are classed as tetranortriterpenes and are abundant in sweet or sour-scented citrus fruit and other plants of the families Cucurbitaceae, Rutaceae, and Meliaceae. Cedrelone exhibits antifeedant activity, it has insecticidal activity and its derivatives exert antimicrobial activities, some show good antifungal activity.
Obacunone, a natural limoniod, is an active component of the herbs Dictamnus dasycarpus and Phellodendron amurense used in the Chinese pharmacopy. Owing to its multiple health benefits, several studies have investigated its multi-targeting potential action mechanisms. A latge survey has indicated that it has great potential to become a promising therapeutic option against tumors, fibrotic diseases, bone and cholesterol metabolism diseases, and infections of pathogenic microorganisms, among others (Zhou Y et al., Molecules 2024, 29(8), 1791).
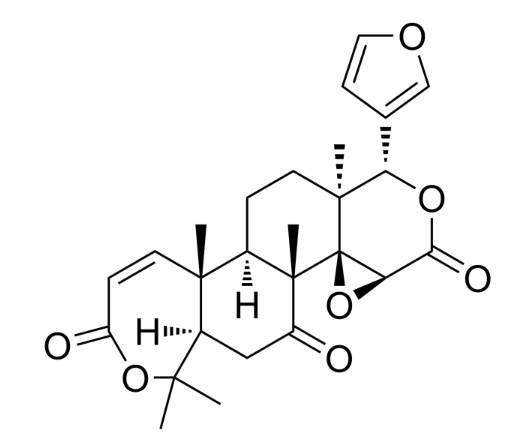 Obacunone
Obacunone
Quassin is a white bitter, crystalline substance that is the prototypical example of the family of quassinoids. More than 200 currently-known quassinoids have been isolated and identified from various species of Simaroubaceae family. Quassin has been extracted in 1937 from Quassia amara, a tropical tree. It is one of the most bitter substances found in nature, with a bitter threshold of 0.08 ppm. It is used as additives in soft drinks and cosmetics. It has insecticidal and insect repellent properties that are used in some traditional medicines. Although its skeleton possesses 20 carbon atoms, quassin is not a diterpene but rather a triterpene lactone, which derives from euphol by loss of 10 carbon atoms including C4.
Due to their distinctive structural features, quassinoids commonly exhibit diverse bioactivities, including insecticidal, antifeedant activities (Xin B et al., J Agric Food Chem 03 Nov 2025), antitumor, and anti-inflammatory properties.
The names, classes, molecular formula, plant sources and pharmacological activities of quassinoids have been reviewed (Duan ZK et al., Phytochemistry 2021, 187, 112769).
Dammarane is a tetracyclic triterpene found in sapogenins like those of ginseng (ginsenosides: panaxatriol and protopanaxadiol). Compounds of the series were first isolated from dammar resin, a natural resin from a tropical tree Shorea javanica (Dipterocarpaceae). Dammarane-type triterpenoids began to draw attention when several publications showed their anticancer activities toward a variety of cancers. Protopanaxadiol is an aglycone of ginsenosides. It is found in ginseng (Panax ginseng) and in notoginseng (Panax pseudoginseng). Several observations suggested that this compound can induce apoptosis in HeLa cells, highlighting its potential as a novel natural therapeutic for cervical cancer (Lu R et al., Food Biosci 9 November 2024, 105388).
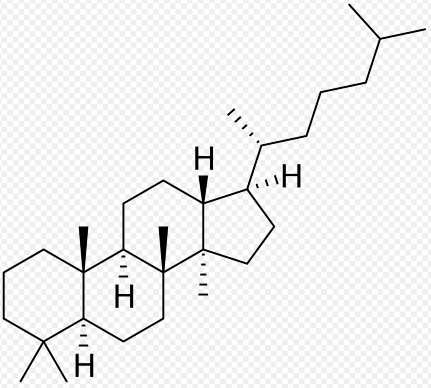
Dammarane
Panaxatriol is an ginsenoside compound, a dammarane-type tetracyclic triterpene sapogenin found in ginseng (Panax ginseng) and in notoginseng (Panax pseudoginseng).
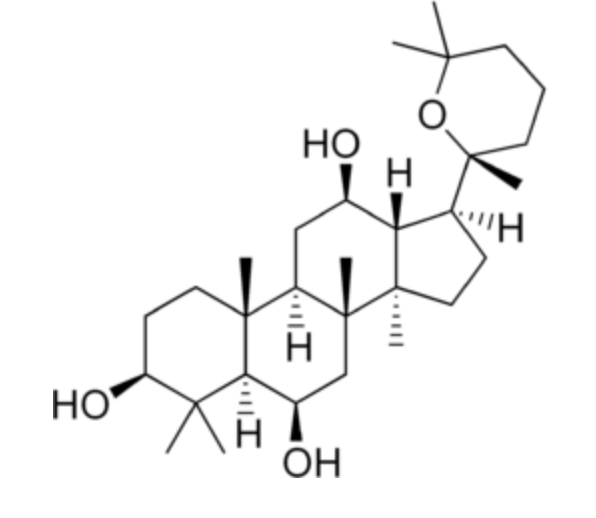 Panaxatriol
Panaxatriol
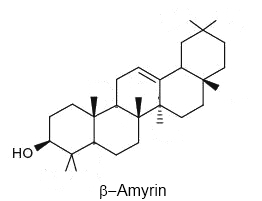
Oleanane is a natural triterpenoid of the oleanoid series, which consists of pentacyclic triterpenoids (such as beta-amyrin and taxerol) where all rings are six-membered. It is commonly found in woody angiosperms and as a result is often used as an indicator of these plants in the fossil record. It must be recalled that contact with seawater during early sedimentation processes can increase the concentration of oleananes in the mature sediment.
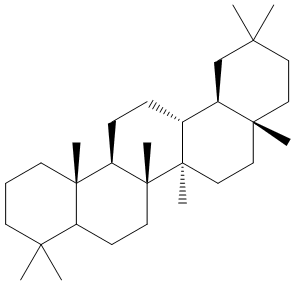 Oleanane
Oleanane
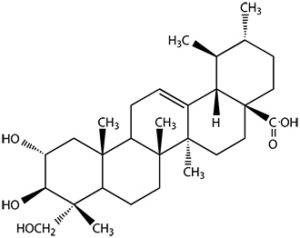
Asiatic acid
Boswellic acids are a series of pentacyclic terpenoid molecules that are produced by plants in the genus Boswellia. These compounds appear in the resin of the plant; it is estimated that they make up 30% of the resin of Boswellia serrata. This resin is an ancient drug prescribed in the Indian traditional medicine (Ayurveda) for treatment of coughs, colds, hoarseness, bronchitis, asthma, dyspnea and diarrhea. Current research suggests it also has therapeutic potential in modern medical practice (Vuddanda PR et al., J Herbal Med 2016, 6, 163).
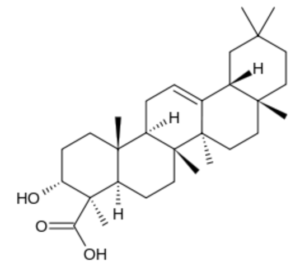
α-Boswellic acid
Acetyl-alpha-boswellic acid and acetyl-beta-boswellic acid have a hydroxyl group instead of an acetyl group. Beta-boswellic acid, keto-beta-boswellic acid, and acetyl-keto-beta-boswellic acid (AKBA) have been indicated in apoptosis of cancer cells. Acetyl-boswellic acids also exhibit anti-inflammatory behaviour by inhibiting the enzyme 5-lipoxygenase.
Acetyl-11-Keto-β-boswellic Acid has been involved in the acceleration of the repair of spinal cord injury in rats thus providing a basis for the development of that terpene as a clinical treatment for spinal cord injury (Wang Y et al., Int J Mol Sci 2024, 25, 358).
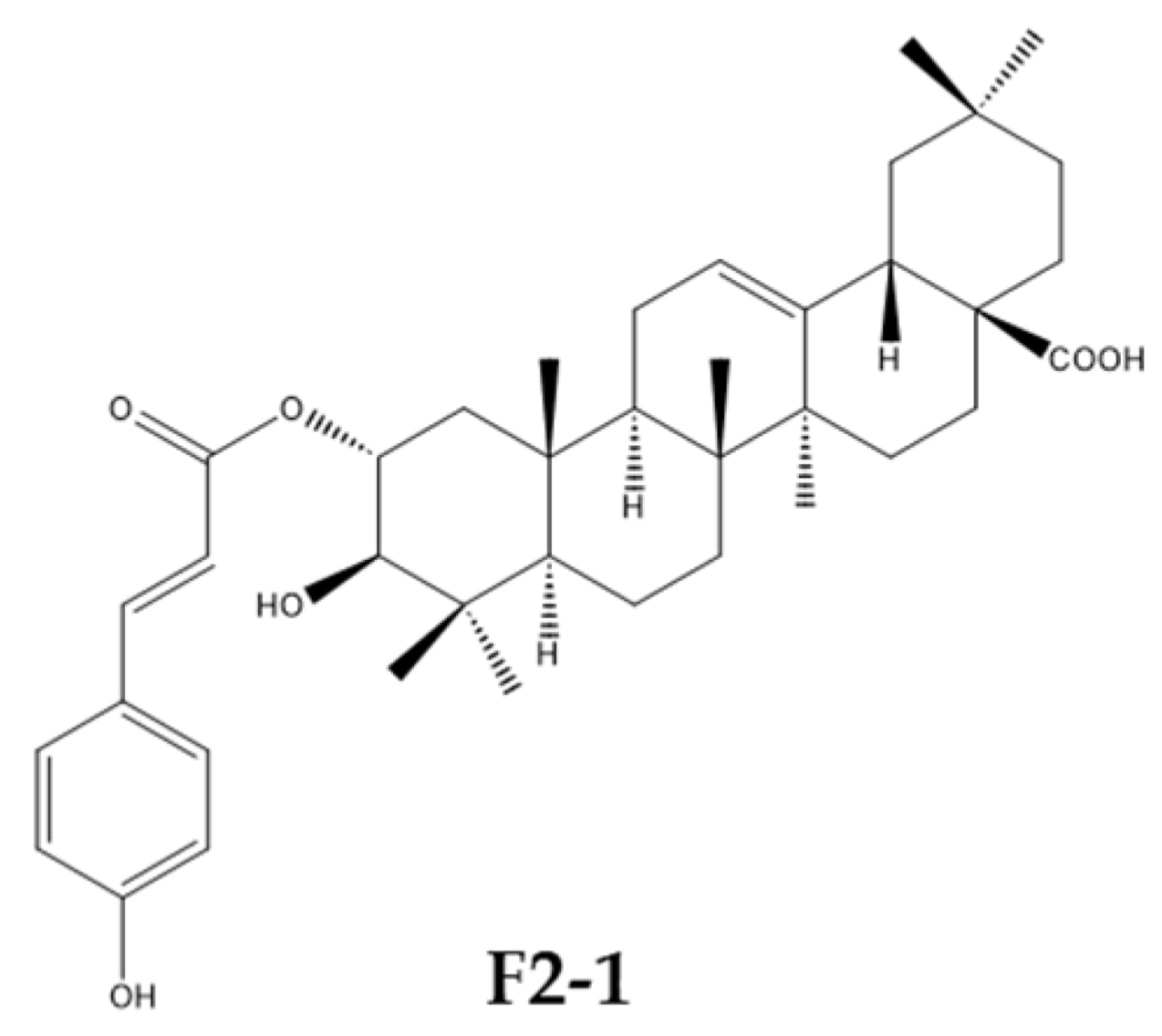
Cinnamoyl triterpenoids are natural compounds isolated from several plants. Some of them have been extracted from plants adapted to high-salt environments (Hippophae rhamnoides) and were shown to be cytotoxic and have antiviral effectiveness against coronaviruses HCoV-229E and SARS-CoV-2 (Al Ibrahim M et al., Int J Mol Sci 2023, 24, 16617). 2-O-trans-p-coumaroyl-maslinic acid (F2-1) was one of the most active compounds. These results suggest that Hippophae rhamnoides could represent a source of potential antiviral agents against human coronaviruses.
Arborinol, is a pentacyclic triterpenoid extracted for the first time from the shrub Glycosmis arborea, but it remains after a plant decomposes. It is often converted to arborane, compound detectable in rocks. Arborane was seen as a marker for the existence of flowering plants in ancient rocks but the discovery that bacteria can also synthesise arborane molecules means that arborane is not a reliable marker for flowering plants. Indeed, it has been detected in rocks which pre-date the origin of plants.
Arborinol
Serratane-type compounds constitute an important group of pentacyclic triterpenes with an unusual seven carbons ring. They are synthesized through the cyclisation of bis-epoxy-squalene and not from epoxy-squalene, the precursor of more common pentacyclic triterpenes (Xu R et al., Phytochemistry 2004, 65, 261).
More than 100 different serratenoids (serratenes and derivatives have been reported, most having been identified in club mosses, conifers, and in few angiosperms (Primulaceae and Leguminosae).
Serratene
As opposed to other serratenoids, methoxy-serratenes appear constrained to conifers, and more specifically to Pinaceae (mainly Pinus, Picea) (Le Milbeau C et al., Org Geochem 2013, 55, 45). In addition to diterpenoids, they potentially constitute a new powerful tool for palaeoenvironmental reconstruction.
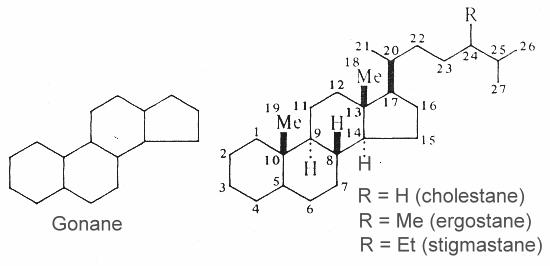
Steroids are modified triterpenes which derived also from squalene by cyclization, unsaturation and substitution. The nucleus of all steroids is the tetracyclic C17 hydrocarbon 1,2-cyclopentanoperhydrophenanthrene (gonane or sterane) substituted by methyl groups at C10 and C13, as well as an alkyl side-chain at C17. Steroids may possess a nucleus derived from the former one by one or more C-C bond scissions or ring expansions or contractions.
Gonane and three examples of basic unsubstituted steroids are shown below.
Unsaturated steroids with most of the skeleton of cholestane containing a 3b-hydroxyl group and an aliphatic side chain of 8 or more carbon atoms attached to position 17 form the group of sterols.
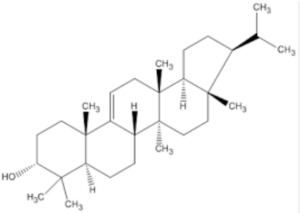
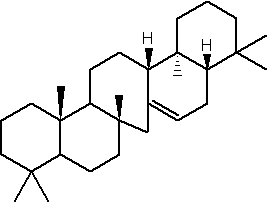
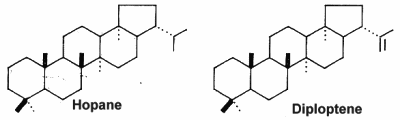
The hopanoids are pentacyclic triterpenoids based on the hopane skeleton (with a five-membered ring E) are widely distributed in prokaryotes but were not detected in Archaea (Rohmer M et al., J Gen Microbiol 1984, 130, 1137). They are often considered bacterial sterol analogs and were first isolated from resins of tropical trees of the genus Hopea by the Ourisson’s team. In most cases, the hopanoid content of the cell is comparable with the cholesterol content of eukaryotic cells. They are considered as membrane rigidifiers. Furthermore, they are the precursors of several derived compounds (homohopanoids) in sediments and oils (Ourisson G et al., Pure Appl Chem 1979, 51, 709) and thus could be considered as the most abundant natural products on earth (Ourisson G et al., Accounts Chem Res 1992, 25, 398). Although oxygen is not required for hopanoid biosynthesis, the vast majority of known hopanoid producers are aerobic or micro-aerophilic bacteria (Rohmer M et al., J Gen Microbiol 1984, 130, 1137). Thus, hopanoids are found predominantly in aerobic methanotrophs, heterotrophs and cyanobacteria, but have also been found in some anaerobic bacteria (Sinninghe Damsté JS et al., Org Chem 2004, 35, 561).
The simplest C30 hopanoid is diploptene.
Cyanobacteria are presently the only known bacteria to synthesize abundant 2-methylhopanoids with an extended polyhydroxylated side chain. The most abundant hopanoids are C35 bacteriohopanepolyols in which the side-chain of the parent structure contains a variable number of vicinal hydroxyl groups
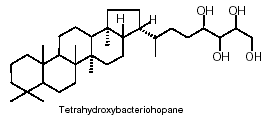
The hopanoids are widely distributed in bacteria and blue-green algae where they are important cell membrane constituents. It is often said that hopanoids are the “most abundant natural products on Earth”.
As hopanoids are very stable and are not easily degraded, they are receiving an intense attention as biological markers with applications for geochemical studies of petroleum source rocks and oils. These biomarkers are mainly derived from bacterial bacteriohopanepolyols (biohopanoids). The 2-methylhopane derivatives are abundant in organic-rich sediments as old as 2,500Myr. These lipids may help constrain the age of the oldest cyanobacteria and the advent of oxygenic photosynthesis. They could also be used to quantify the ecological importance of cyanobacteria through geological time (Summons RE et al., Nature 1999, 400, 554). More recent investigations have shown that the production of substantial quantities of polyhydroxylated 2-methylhopanoids may occur in phototrophs other than cyanobacteria and that their biosynthesis does not require molecular oxygen (Rashby SE et al., PNAS 2007, 104, 15099). Hopanoids, and especially their C-2 methylated derivatives, have been found in high abundance in membranes of several plant symbionts (Bradyrhizobium, Frankia). In these bacteria, they contribute to growth and protection against stresses (Kulkarni G et al., mBio 2015, 6(5):e01251-15). In some symbiotic bacteria, an unusual lipopolysaccharide bearing a hopanoid covalently linked to lipid A has been described (Silipo A et al., Nat Commun 2014, 5:5106). a brief review of the hopanoid biosynthesis may be found in the Sohlenkamp paper (Sohlenkamp C et al., FEMS microbiol Rev 2016, 40, 133).
Aminohopanoids not very different from the previous one have been identified in several methylotrophic bacteria (Neunlist S et al., Biochem J 1985, 231, 635). One of the most abundant is a aminohopane pentol, mixed with other parent compounds (triol and tetrol). This C30 aminopentol is a possible precursor of the widespread C29 hopanoid chemical fossils.
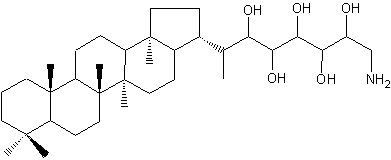
Aminobacteriohopane
Different bacterial groups possess recognizable biohopanoid distributions, giving hopanoids marker potential for specific bacterial populations and environmental conditions. More than 150 individual hopane derivatives have been isolated from various types of sedimentary organic matter.
Other pentacyclic triterpenoids based on the lupane skeleton include a very large number of naturally occurring members with different functional groups which are found in vegetables and fruit. Among them, lupeol is one of the most ubiquitous compounds. It is found in a variety of plants, including mango, Acacia visco, Abronia villosa andBauhinia acuminata (Nakkala K et al., Biomed Chromatogr 2022, 36, e5448). It is also found in dandelion coffee. Lupeol is present as a major component in Camellia japonica leaf.
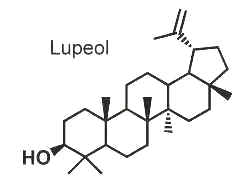
Lupeol was shown to have various pharmacological activities, inducing antiprotozoal, antimicrobial, anti-inflammatory, chemopreventive properties, anti-arthritic responses and inhibition of tumor growth (Saleem M et al., Cancer Res 2005, 65, 11203) in animal cells. Lupeol has shown promising benefits in the management of many other human diseases such as diabetes, obesity, cardiovascular diseases, kidney and liver problems, skin diseases, and neurological disorders. Despite significant progress in molecular pharmacology, the clinical application of lupeol is limited due to poor bioavailability and insufficient knowledge on its mode of action (Sohag AAM et al., Pytomedicine 2022, 99, 154012). Results have demonstrated that Lupeol exhibited significant anti-proliferative effects on HepG2 cells with an IC50 of 42.82 μM at 24 h. Furthermore, Lupeol treatment significantly inhibited cell migration and suppressed the secretion of exosomes (Zhang X et al., Food Sci Nutr 2026, 14, e71654).
It could be an important component of the cholesterol‐lowering activity of miracle fruit (Synsepalum dulcificum) (Wang W et al., J Food Biochem 2020, 44, e13185). The binding and inhibitory potentials of lupeol have been investigated and proved to be effective against certain receptor proteins and enzymes in Altzheimer’s disease studies by computational molecular docking approaches (Park JS et al., Nutrients 2023, 15, 3059).
Long-chain fatty acid esters (C20, C22 and C26) of lupeol extracted from an African plant, Holarrhena floribunda (Apocynaceae), were shown to have strong antimalarial activity, especially against drug-resistant strains of Plasmodium falciparum (Fotie J et al., J Nat Prod 2006, 69, 62).
Fatty acid esters (palmitic or stearic acid) of lupeol have been isolated from green propolis produced by honeybees from vegetative apices of the Asteraceae Baccharis dracunculifolia from Brazil (Furukawa S et al., Chem Pharm Bull 2002, 50, 439). These compounds were named procrim a and b.
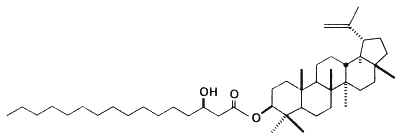
Palmitic acid ester of lupeol (Procrim a)
Betulin, a pentacyclic triterpene is present in large quantities in the outer bark of birches (Betula, Betulaceae). A derivative, betulinic acid, is also abundantly present in nature.
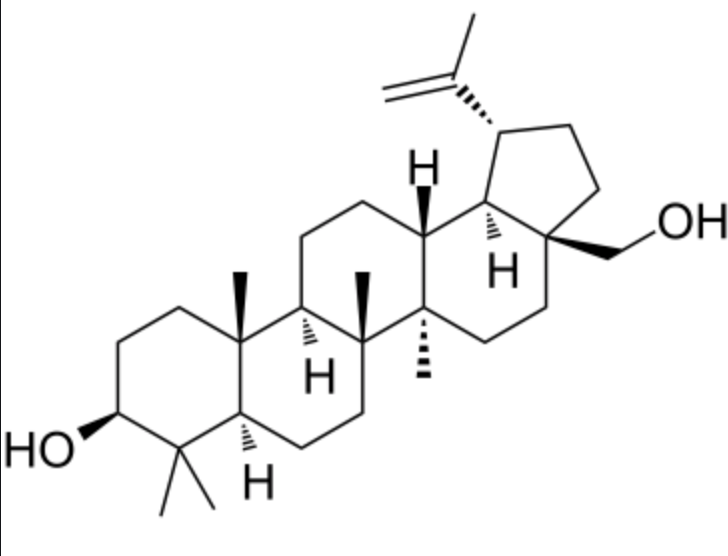
Betulin
It is commonly isolated from the bark of birch trees where it forms up to 30% of the dry weight. This compound gives the tree its white color which likely protect the tree from overheating by the sun. As a result, birches are some of the northern most occurring deciduous trees. It can be also obtained from more than two hundred plants species, although the richest source is the Betulaceae family, especially Betula alba, B. pubescens, B. platyphylla and B. pendula. It has been demonstrated that white birch species’ bark contains betulin among other compounds that likely make the bark unprofitable for herbivores. Thus, the white bark has features consistent with a visual signal for mammalian herbivores and could act as a visual aposematic signal of chemical defence (Ireland H M et al., Plant Ecology & Diversity 2022, 15, 93).
Betulin shows a wide spectrum of biological and pharmacological properties, such as anti-HIV, anti-inflammatory, and, considered the most important, anti-cancer. Although its specific mechanism of action against malignant cells is still in debate, the activity of betulin acid has been linked to the induction of the intrinsic pathway of apoptosis. As this process occurs with the sparing of non-cancer cells, and the induction of apoptosis can occur under conditions in which standard therapies fail, both substances seem as promising experimental anti-cancer drugs.
The discovery, structure, organic synthesis and derivatives of betulin, and the potential molecular mechanisms of action and numerous medical applications of betulin and betulinic acid have been reviewed (
Ursolic acid is present in numerous plants and is the best known triterpene from the ursane group. It amounts at more than 1 g per 100 g dry weight in Thymus, Rosmarinus, Salvia, Lavandula, and Eucalyptus. It is found at high concentration in coffee seeds and in apple fruit (mainly in apple peels) and in fruit of loquat (Eriobotrya japonica). It is used in cosmetics as an anti-inflammatory, antibacterial and antifungal drug. Ursolic acid is said to inhibit some form of cancer, particularly multiple myeloma (Pathak AK et al., Mol Cancer Res 2007, 5, 943) and colorectal cancer. It has been found that ursolic acid may inhibit the malignant phenotype, induce apoptosis, and arrest the cancer cell cycle by attenuating the Wnt/β-catenin signaling axis, providing insights into the mechanism for the potency of UA on CRC (Zhao H et al., J Agric Food Chem 2023, 71, 3981).
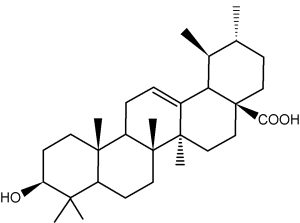
Ursolic acid
Corosolic acid is is found in Lagerstroemia speciosa andEriobotrya japonica. It is similar in structure to ursolic acid, differing only in the fact that it has a 2-alpha-hydroxy attachment (Wikipedia). Investigations have demonstrated that this triterpene could be useful for the treatment of cardiovascular diseases and other diseases of a similar nature (Alkholifi FK et al., Molecules 2023, 28, 929). It could also improve the symptoms that are associated with the metabolic syndrome via multiple mechanisms. It is emerging as a promising candidate for the development of next-generation antiosteoporotic agents (Zhao C et al., Free Rad Biol Med 2025, 241, 882).
A compound very similar to ursolic acid, oleanolic acid, is widely distributed in food and medicinal plants.
It is hepatoprotective and exhibits antitumor and antiviral properties. It is the main constituent of grape berry cuticular waxes (Szakiel A et al., Phytochem Rev 2012, 11, 263). Its presence could be at the origin of the numerous health benefits, including protection against heart disease, ascribed to the moderate consumption of wine. Oleanolic acid and another parent compounds, maslinic acid, are concentrated in olive cuticular waxes, representing 31–44 % and 55–68 % of total wax extract, respectively. These compounds could be related to the numerous health-promoting properties, such as anticancer, antihyperglycemic and antiparasitic activities, reported for olive consumption (Szakiel A et al., Phytochem Rev 2012, 11, 263).
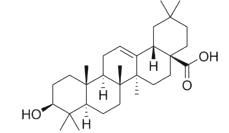
Oleanolic acid
A parent compound of oleanolic acid (oleanonic acid), with a ketone group instead of the hydroxyl group, is present in Pistachia resin and has been used to establish that this substance was used in the preparation of pharaonic meat mummies (Clark KA et al., PNAS 2013, 110, 20392).
Another parent compound of oleanolic acid (taraxerol) is an oleanan-3-ol with an alpha-methyl substituent at position 13, a missing methyl group at position 14, and a double bond between 14 and 15. It was found in various higher plants of various families, including Taraxacum officinale (Asteraceae). It is also named “skimmiol“. A large number of medicinal plants are known to have this compound in their leaves, roots or seed oil. Taraxerol is present in very high levels in many species of mangrove tree leaves, e.g. Rhizophora mangle (red mangrove) and Rhizophora racemosa. Therefore, it is used in various studies as a proxy for mangrove input (Sefton JP et al., Organ Geochem 2024, 191, 104767).
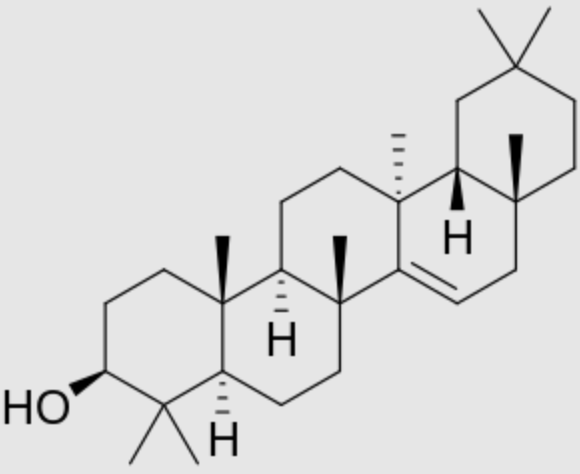 Taraxerol
Taraxerol
Taraxasterol has a structure similar to taraxerol, it is one of the bioactive triterpenoids found in dandelion (Asteraceae). Taraxasterol has been shown to have significant preventive and therapeutic effects in the animal or cellular models, including liver damage, gastritis, colitis, arthritis, pneumonia, tumors, and immune system diseases (review in: Jiao F et al., Front Pharmacol 04 August 2022, 13). A growing number of research presents the novel and potential effect of dandelion in treating or preventing cancers, including breast cancer, gastric cancer, colorectal cancer, and other malignant tumors (Wang A et al., Nutrients 2025, 17, 3769; Ovadje P et al., Oncotarget. 2016, 7, 73080).
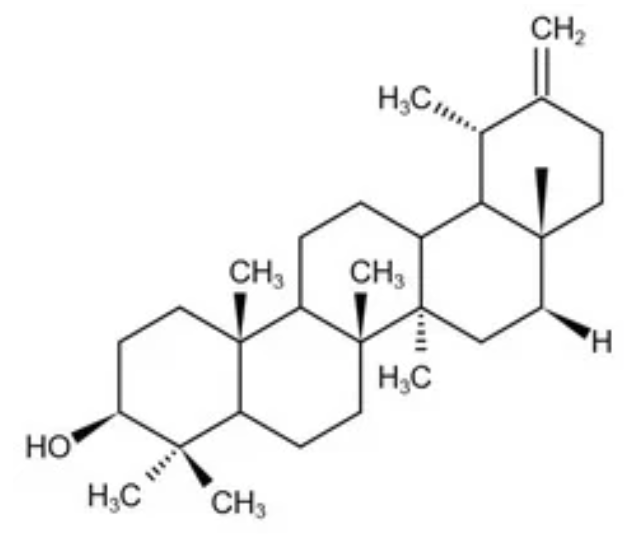 Taraxasterol
Taraxasterol
As ursolic acid but in contrast with the hopane series, which possesses a five-membered ring E, the gammacerane skeleton is characterized by a six-membered ring (see below).
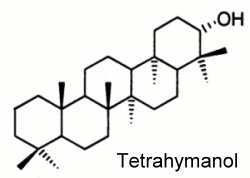
Tetrahymanol (gammaceran-21a-ol), a typical representative of the series, was first isolated from the ciliate protozoan Tetrahymena pyriformis (Mallory FB et al., J Am Chem Soc 1963, 85, 1362). Later, it was detected in a number of other eukaryotes, e.g. in ferns, fungi and some other ciliates. Its occurrence was long thought to be restricted to eukaryotes but its presence in sediments pointed out a much more widespread distribution in living organisms. The finding of tetrahymanol in the purple nonsulfur bacterium Rhodopseudomonas palustris opened new insights into the biochemistry of these molecules in bacteria (Kleemann G et al., J Gen Microbiol 1990, 136, 2551). Tetrahymanol and novel methylated homologues were discovered in nitrogen-fixing bacterium Bradyrhizobium japonicum (Bravo JM et al., Eur J Biochem 2001, 268, 1323).
Gammacerane structures were shown to be reliable geochemical indicators for water column stratification in marine or in lacustrine deposits (Sinninghe Damste JS et al., Geochim Cosmochim Acta 1995, 59, 1895).
Celastrol is a hopanoid triterpene extracted from a plant used in the Chinese medicine, Tripterygium wilfordii. That plant is useful to treat inflammatory and autoimmune diseases. New research have demonstrated that it is a potent proteasome inhibitor and can help in the treatment of prostate cancer (Yang H et al., Cancer Res 2006, 66, 4758). Several biochemical properties bring about several investigations in the field of cancer treatment or prevention. The antitumor potential of celastrol and the reduction of its toxicity have been extensively reviewed (Jiang Q et al., Biomolecules 2026, 16(5), 620).
Its antioxidant and anti-inflammatory properties has been the basis of research on diseases induced by monocytes and macrophages activation, including neurodegenerative process (Allison AC et al., Prog Neuropsychopharmacol Biol Psychiatry 2001, 25, 1341).
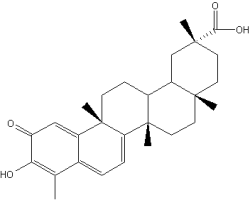
Celastrol
Triterpene dimers is a small group of compounds found in nature (from plants precisely). Until 2021, the chemical and pharmacological works reported in the literature indicated the identification of 90 natural dimeric triterpenes and 11 synthetic derivatives from 19 plants species. Very few of them have been biologically evaluated for their antibacterial, antioxidant, antiproliferative or molluscicide activities. A review had compiled the literature on the occurrence, chemistry and biological activities of the triterpenoid dimers (Happi GM et al., Phytochemistry 2022, 200, 113242).
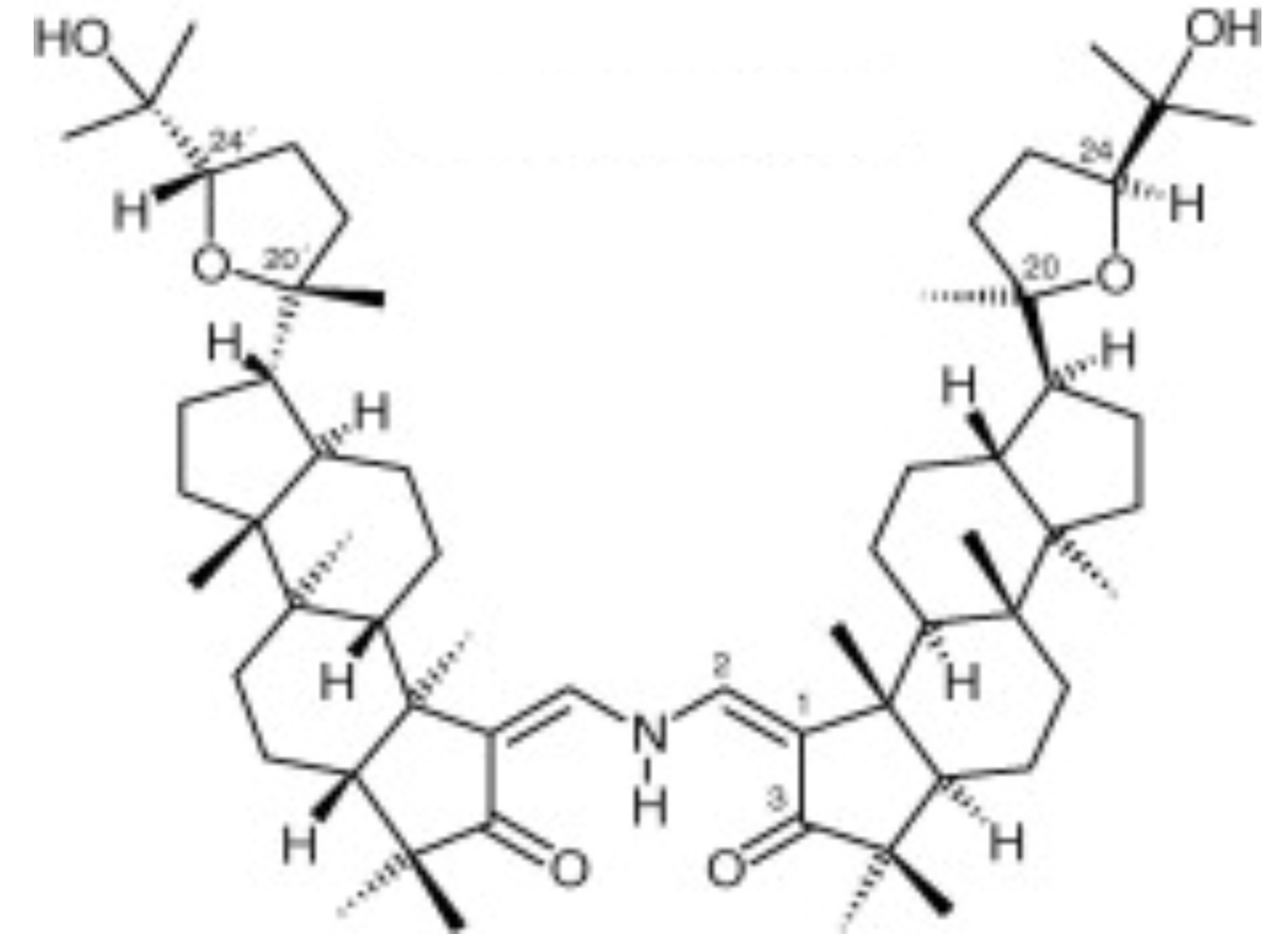
Silvaglenamin
DISPERSIVE LIQUID-LIQUID MICROEXTRACTION
Lire la suiteDevenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire