
THIN-LAYER CHROMATOGRAPHY
Thin-layer chromatography (TLC) is perhaps the most efficient and versatile technique ever discovered for separation of complex lipid mixtures as well as for individual lipids. TLC is rapid since up to 10 spots may be run frequently in less than an hour, it does not require any special equipment and allows the characterization of the separated lipids with various staining reactions. Fractions can be easily recovered by elution and studied by other procedures.
We give below the simplest recipe to separate rapidly a complex lipid mixture in two fractions: neutral lipids and polar lipids (phospholipids and glycolipids).
Apparatus:
Commercial TLC plates (Silica gel G 60, Merck, Ref. 5721), 20 x 20 cm, to be cut at the chosen width with a glass cutter.
Hamilton micro-syringes, 10 or 50 µl.
Scrapers with acute blades (Alltech).
All-glass tank with ground glass cover.
Reagents
Diethyl ether, methanol, acetic acid, chloroform, hexane
Eluant mixture: diethyl ether/methanol/acetic acid (90/1/1, v/v)
Primuline spray (5 mg in 100 ml of acetone/water (80/20, v/v)
Procedure
New plates are pre-run in a special tank containing chloroform/methanol (50/50, v/v) up to the top to remove any contaminant from the silica gel. Clean plates are stored in another glass tank.
Concentrate solutions of lipids in chloroform/methanol mixtures (2/1, v/v) are applied (1 to 10 µl per spot) as fine rows at 2.5 cm of the bottom of the plate and separated from each other by 1 cm. After evaporation with the help of a nitrogen flow or a cool air blowing (air dryer), plates are placed immediately in the elution tank (height of solvent: 5 mm) and the solvent is allowed to ascend to within about 2 cm of the top of the plates. It is possible to allow only a 10 cm migration for this separation. Remove from the tank and allow the solvent to evaporate in the fume hood.
When one smells only slightly acetic acid, the primuline solution is sprayed with an all-glass atomizer and the plate is viewed under ultraviolet light (340 nm). Lipids appear as bright yellow spots on a clear background, neutral lipids are at a low distance from the solvent front and polar lipids remain at the depot line.
Lipids are recovered from the silica gel in eluting the spots in glass tubes by:
– for neutral lipids: 2 x 3 ml hexane/diethyl ether (50/50, v/v) or 2 x 3 ml chloroform/methanol/diethyl ether (10/10/10, v/v).
– for polar lipids: 2 x 5 ml chloroform/methanol/water (5/5/1, v/v) and addition of 5 ml water to the combined eluates (10 ml). After vortexing 1 min and a short centrifugation, polar lipids are recovered in the lower phase (chloroform).
This fraction can be used to determine the phospholipid (via the amount of phosphorus or fatty acids) or the glycolipid content (via the amount of sugar).
Comments :
Primuline is a fluorescent dye being a thiazole very soluble in water and ethanol. It is also known as “Direct yellow 7” and its CI number is 49010.
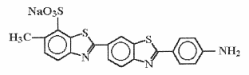
Primuline may be found in the catalogues of Aldrich Chemical or Conn’s Biological Stains.
DISPERSIVE LIQUID-LIQUID MICROEXTRACTION
Lire la suiteDevenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire