
They contain a hydrophobic ceramide moiety and a hydrophilic moiety which consists of mono- or oligosaccharide unit bound to the C-1 hydroxy group of the ceramide. Generally, the terminal galactose residue is esterified with sulfate at carbon 3. Details on their structure and metabolism may be found in the Farooqui’s review (Adv Lipid Res 1981, 18, 159).
Three major sulfosphingolipids are known from mammalian tissues :
– cerebroside 3-sulfate or sulfatide is a sulfogalactosyl ceramide
– ceramide dihexosyl sulfate is a sulfolactosyl ceramide
– ceramide tetrahexosyl sulfate
![]()
This group (known also as cerebroside 3-sulfate) is mainly formed of 3-sulfate esters of galactosylcerebrosides (galactosyl-3-sulfate esters) and is found in mammalian tissues as the corresponding cerebroside group.
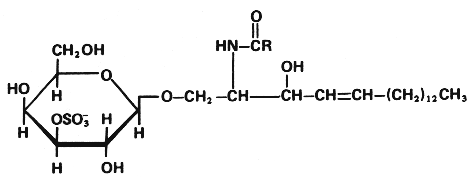
Crebroside sulfate (sulfatide)
(R = fatty acid carbon chain)
The first evidence for the existence of sulfur containing lipids was given by Thudichum (1884) who isolated this lipid from human brain and gave it the name sulfatide. The first complete description of the structure was given by Blix (Z Physiol Chem 1933, 219, 82).
Sulfatide is a major constituent of brain lipids and is found in trace amount in other tissues. It is an essential glycosphingolipid in the peripheral as well as the central nervous system, constituting 4-6 mole% of the total lipids in adult brain myelin. Sulfatide is also found in kidney and retina. About half the fatty acids are a-hydroxylated.
This compound is formed from a cerebroside molecule by a sulfotransferase reaction with a donor nucleotide, 3′-phosphoadenosine-5’phosphosulfate. This enzyme is very active during the myelinisation process but the turnover of the sulfate group is extremely slow. Curiously, these sulfatides are also found in tissues that are very active in sodium transport (kidney, salt glands, gills).
Sulfatide mediates diverse biological processes including the regulation of cell growth , protein trafficking, signal transduction, cell adhesion, neuronal plasticity and morphogenesis (Ishizuka I, Prog Lipid Res 1997, 36, 245). Experiments on mice lacking sulfotransferase demonstrated that sulfatide is required for myelinisation and in preventing neuronal degenerescence, abnormalities in paranodal junctions (Honke K et al., PNAS 2002, 4227), and maintenance of ion channels (Ishibashi T et al., J Neurosci 2002, 22, 6507).
The analysis of human brain tissues demonstrates that a marked decrease in sulfatides is associated with Altzheimer disease pathology even in subjects with very mild dementia, these changes may be linked with early events in the pathological process (Han X et al., J Neurochem 2002, 82, 809).
Sulfatides were shown to be about three times more concentrated in the kidneys of marine mammals than in those of terrestrial ones (Nagai KI et al., Glycoconj J 2008, 25, 723). These differences suggest that these acidic compounds may contribute to the maintainance of osmotic balance of their body fluid agaiinst sea water.
A sulfatide with two hexose moieties (glucose and galactose) has been isolated from human kidney (Martensson E, Acta Chem Scand 1963, 17, 1174). Equimolar amounts of ceramide, glucose, galactose, and sulfate are found in the molecule. The sulfate group is esterified, as in sulfatides, with carbon 3 of the galactose moiety. The linkage between galactose and glucose is identical to lactose (b-1-4). Its fatty acid composition is similar to that of sulfatides.
Lactosyl sulfatides were shown to be about three times more concentrated in the kidneys of marine mammals than in those of terrestrial ones (Nagai KI et al., Glycoconj J 2008, 25, 723). These differences suggest that these acidic compounds may contribute to the maintainance of osmotic balance of their body fluid agaiinst sea water.
This sulfated lipid was discovered by Slomiany BL in hog gastric mucosa (Biochim Biophys Acta 1974, 348, 388). The oligosaccharide group is composed of one galactose and two glucose moieties. The sulfate group is esterified, as in the other sulfosphingolipids, with carbon 3 of the galactose moiety. It appears that the fatty acids are poorly hydroxylated.
A sulfoglycosphingolipid with an oligosaccharide chain composed of four units was characterized as the major sulfoglycolipid of mouse small intestine (Leffler H et al., J Biol Chem 1986, 361, 1440). Its global formula may be written as follow :
–O3SO-3Gal1-3GalNAc1-4Gal1-4Glc1-1Cer
This sulfoglycolipid is enriched in epithelial cells of mouse small intestine where it constitutes at least 90% of the acidic glycolipids and 4-8% of the total glycosphingolipids.
DISPERSIVE LIQUID-LIQUID MICROEXTRACTION
Lire la suiteDevenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire