
Fatty acid hydroperoxides generated by plant lipoxygenases from linoleic and linolenic acids are known to serve as substrates for a divinyl ether synthase which produces divinyl ether fatty acids. Up to date divinyl ethers were detected only within the plant kingdom.
The discovery of that class of compounds dates back to 1972, when Galliard T et al. described the structures of two ether C18 fatty acids generated by homogenates of the potato tuber (Galliard T et al., Biochem J 1972, 129, 743). These compounds, named colneleic acid (from linoleic acid) and colnelenic acid (from linolenic acid), could be also produced in potato leaves and tomato roots by rearrangement of 9-hydroperoxides.
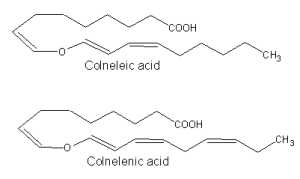
Isomers of colneleic and colnelenic acids were isolated from homogenates of leaves of Clematis vitalba (Ranunculaceae) (Hamberg M, Lipids 2004, 39, 565).
Similarly, 13-lipoxygenase-generated hydroperoxides serve as precursor of other divinyl ether fatty acids which are produced in bulbs of garlic (Grechkin AN et al., FEBS Lett 1995, 371, 159) or Ranunculus leaves (Hamberg M, Lipids 1998, 33, 1061). These compounds were named etheroleic and etherolenic acids.
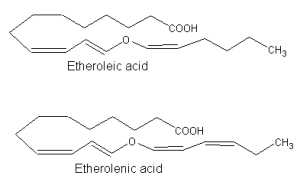
The physiological significance of divinyl ethers is still not fully studied. As infection of potato leaves leads to increased levels of divinyl ether synthase, it was suggested that this pathway could be of importance in the defense of plants against attacking pathogens (Göbel C et al., Biochim Biophys Acta 2002, 1584, 55).
Similar structures have been discovered in the brown alga Laminaria sinclairii, with 18 or 20 carbons and 4, 5 or 6 double bonds (Proteau PJ et al., Lipids 1993, 28, 783), and in the red alga Polyneura latissima, with 20 carbons and 5 double bonds (Jiang ZD et al., Lipids 1997, 32, 231).
DISPERSIVE LIQUID-LIQUID MICROEXTRACTION
Lire la suiteDevenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire