
Although the dicarboxylic acids do not occur in appreciable amounts as components of animal or vegetal lipids, they are in general important metabolic products of fatty acids since they originate from them by oxidation. Dicarboxylic acids are suitable substrates for preparation of organic acids for the pharmaceutical and food industries. Furthermore, they are useful materials for the preparation of fragrances, polyamides, adhesives, lubricants, and polyesters.
They have the general type formula:
HOOC-(CH2)n-COOH
In vegetal, a great variety of molecular forms of dicarboxylic acids are found :
1- simple forms with a straight carbon chain or a branched chain
2- complex forms with a dicarboxylic acid and an alkyl side chain : alkylitaconates
![]()
1 – Simple forms of dicarboxylic acids
Short-chain dicarboxylic acids are of great importance in the general metabolism and up to n=3 they cannot be considered as lipids since their water solubility is important. The simplest of these intermediates is oxalic acid (n=0), the others are malonic (n=1), succinic (n=2) and glutaric (n=3) acids.
The other lipid members of the group found in natural products or from synthesis have a “n” value from 4 up to 21.
Adipic acid (n=4) : Despite its name (in Latin adipis is fat), this acid (hexanedioic acid) is not a normal constituent of natural lipids but is a product of oxidative rancidity (lipid peroxidation). It was obtained by Dieterle W et al. (Ber. 1884, 17, 2221) by oxidation of castor oil with nitric acid (splitting of the carbon chain close to the OH group). Synthesized in 1902 from tetramethylene bromide, it is now obtained by oxidation of cyclohexanol or cyclohexane. It has several industrial uses in the production of adhesives, plasticizers, gelatinizing agents, hydraulic fluids, lubricants, emollients, as an additive in the manufacture of some form of nylon (nylon-6,6), polyurethane foams, leather tanning, urethane and also as an acidulant in foods. Adipic acid is used after esterification with various groups such as dicapryl, di(ethylhexyl), diisobutyl, and diisodecyl.
A graphic chart describing the biosynthesis of adipic acid via omega oxidation may be found on the BioCarta web site.
Pimelic acid (n=5) : this acid (heptanedioic acid), from the Greek pimelh (pimele fat), as adipic acid, was isolated from oxidized fats. It was obtained in 1884 by Ganttner F et al. (Ber. 1884, 17, 2212) as a product of ricinoleic acid (hydroxylated oleic acid) from castor oil.
Suberic acid (n=6) : it was firstly produced by nitric acid oxidation of cork (Latin suber) material and then from castor oil (Tilley TG, Ann 1841, 39, 160). The oxidation of ricinoleic acid produces, by splitting at the level of the double bond and at the level of the OH group, at the same time, suberic acid (octanedioic acid) and the next homologue azelaic acid.
Suberic acid was used in the manufacture of alkyd resins and in the synthesis of polyamides leading to nylon.
Azelaic acid (n=7) : nonanedioic acid is the best known dicarboxylic acid. Its name stems from the action of nitric acid (azote, nitrogen, or azotic, nitric) oxidation of oleic or elaidic acid. It was detected among products of rancid fats (Nicolet BH et al., J Ind Eng Chem 1916, 8, 416 and Nunn L et al., Biochem J 1938, 32, 1974). Its origin explains for its presence in poorly preserved samples of linseed oil and in specimens of ointment removed from Egyptian tombs 5000 years old (Banks A et al., Analyst 1933, 58, 265). Azelaic acid was prepared by oxidation of oleic acid with potassium permanganate (Ganttner F et al., Ber. 1881, 14, 1545), but now by oxidative cleavage of oleic acid with chromic acid or by ozonolysis.
Azelaic acid is used, as simple esters or branched-chain esters) in the manufacture of plasticizers (for vinyl chloride resins, rubber), lubricants and greases. Azelaic acid is now used in cosmetics (treatment of acne). It displays bacteriostatic and bactericidal properties against a variety of aerobic and anaerobic micro-organisms present on acne-bearing skin. Azelaic acid was identified as a molecule that accumulated at elevated levels in some parts of plants and was shown to be able to enhance the resistance of plants to infections (Jung HW et al., Science 2009, 324, 89). It was shown that pathogen-induced accumulation of nitric oxide and reactive oxygen species promotes the production of azelaic acid that primes plants for salicylic acid-dependent defences (Wang C et al., Cell Rep 2014, 7:348). The oxidation leads to the formation of oxylipins, which participate in a myriad of signalling pathways.
Currently, ozonolysis is the sole industrial process for the sustainable synthesis of azelaic acid, predominately utilizing oleic acid. It was shown that a new chemical process less harmful for the environment was able to synthesize azelaic acid in 48–72% yields using a non-edible Triadica sebifera seed oil and some other vegetable oils (Sood A et al., Indus Crops Prod 2022, 186, 115139).
Sebacic acid (n=8) : decanedioic acid was named by Thenard LJ (1802) from the Latin sebaceus (tallow candle) or sebum (tallow) in reference to its use in the manufacture of candles. Thenard LJ isolated this compound from distillation products of beef tallow. In 1954, it was reported that it was produced in excess of 10,000 tons annually by alkali fission of castor oil (Kadesch RG, J Am Oil Chem Soc 1954, 31, 568).
Sebacic acid and its derivatives, as azelaic acid, have a variety of industrial uses as plasticizers, lubricants, diffusion pump oils, cosmetics, candles, etc. It is also used in the synthesis of polyamide, as nylon, and of alkyd resins.
An isomer, isosebacic acid, has several applications in the manufacture of vinyl resin plasticizers, extrusion plastics, adhesives, ester lubricants, polyesters, polyurethane resins and synthetic rubber.
Dodecanedioic acid (n=10) : that acid is used in the production of nylon (nylon-6,12), polyamides, coatings, adhesives, greases, polyesters, dyestuffs, detergents, flame retardants, and fragrances. It is now produced by fermentation of long-chain alkanes with a specific strain of Candida tropicalis (Kroha K, Inform 2004, 15, 568). Its monounsaturated analogue is traumatic acid (see below).
Suberic acid, nonanedioic acid (and its mono-unsaturated derivative: 2-nonenedioic acid), decanoic acid (and its mono-unsaturated derivative: 2-decenedioic acid) are present in honey (Schievano et al., J Agric Food Chem 2013, 61, 1747) and are of special interest because they have long been recognized as part of the pheromone system of the honeybee Apis mellifera (Lercker G et al., Lipids 1981, 16, 912). They are produced in the mandibular glands of the queen and the worker honeybees, they regulate their activities in the hive.
It was shown that all these dicarboxylic acids are formed during the drying process of paint oils and that the determination of these decomposition products may be of value in determining the age of old samples.
The higher weight dicarboxylic acids (n=10 to 21) are found in different plant lipids, particularly in what was named erroneously Japan wax (triglycerides containing C20, 21, 22 and 23 dicarboxylic acids besides normal fatty acids) from the sumach tree (Rhus sp.).
Among them, Thapsic acid (n=14) was isolated from the dried roots of the Mediterranean “deadly carrot”, Thapsia garganica (Umbelliferae), but others, as Brassylic acid (n=11), were prepared chemically from different sources. Brassylic acid can be produced chemically from erucic acid by ozonolysis but also by microorganisms (Candida sp) from tridecane. This diacid is produced on a small commercial scale in Japan for the manufacture of fragrances.
A review on the applications and the industrial biotechnology of these molecules has been released by Kroha K (Inform 2004, 15, 568).
A large survey of the dicarboxylic acids present in Mediterranean nuts revealed unusual components (Dembitsky VM et al., Food Chem 2002, 76, 469). A total of 26 minor acids (from 2 in pecan to 8% in peanut) were determined : 8 species derived from butanedioic acid, likely in relation with photosynthesis, and 18 species with a chain from 5 to 22 carbon atoms.
Higher weight acids (>C20) are found in suberin present at vegetal surfaces (outer bark, root epidermis). C16 to C26 α,ω-dioic acids are considered as diagnostic for suberin. With C18:1 and C18:2, their content amount from 24 to 45% of whole suberin. They are present at low levels (< 5%) in plant cutin, except in Arabidopsis where their content can be higher than 50% (Pollard Met al., Tr Plant Sci 2008, 13, 236).
The first allenic dicarboxylic acid, named glutinic acid (2,3-pentadienedioic acid) was isolated from Alnus glutinosa (Betulaceae) (Hans EA, Berich Deut Chem Ges 1908, 40, 4760).
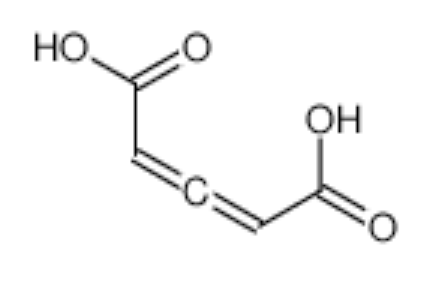
Glutinic acid
It was shown that hyperthermophilic microorganisms specifically contained a large variety of dicarboxylic acids (Carballeira NM et al., J Bacteriol 1997, 179, 2766). This is probably the most important difference between these microorganisms and other marine bacteria. Dioic fatty acids from C16 to C22 were found in an hyperthermophilic archaeon, Pyrococcus furiosus. Short and medium chain (up to 11 carbon atoms) dioic acids have been discovered in Cyanobacteria of the genus Aphanizomenon (Dembitsky VM et al., Biochemistry (Moscow) 2001, 66, 72).
A monounsaturated dicarboxylic acid, traumatic acid, (10E-dodeca-1,12-dicarboxylic acid), was among the first biologically active molecules isolated from plant tissues (English J et al., Science 1939, 90, 329). That dicarboxylic acid was shown to be a potent wound healing agent in plant that stimulates cell division near a wound site (Farmer EE, Plant Mol Biol 1994, 26, 1423), it derives from 18:2 or 18:3 fatty acid hydroperoxides after conversion into oxo fatty acids.
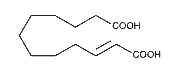
Traumatic acid
While polyunsaturated fatty acids are unusual in plant cuticles, a diunsaturated dicarboxylic acid has been reported as a component of the surface waxes or polyesters of some plant species. Thus, octadeca-c6,c9-diene-1,18-dioate, a derivative of linoleic acid, is present in Arabidopsis and Brassica napus cuticle (Bonaventure G et al., Plant J 2004, 40, 920).
Dicarboxylic acids were shown in 1934 to be produced by ω-oxidation of fatty acids during their catabolism (Verkade PE et al., Biochem J 1934, 28, 31). Thus, these authors have discovered that these compounds appeared in urine after administration of tricaprin and triundecylin. Although the significance of their biosynthesis remains poorly understood, it was demonstrated that ω-oxidation occurs in rat liver but at a low rate, needs oxygen, NADPH and cytochrome P450. It was later shown that this reaction is more important in starving or diabetic animals where 15% of palmitic acid is subjected to w-oxidation and then to β-oxidation (Wada F et al. Biochim Biophys Acta 1977, 487, 261), this generates malonyl-coA which is further used in saturated fatty acid synthesis.
It was proposed recently that dicarboxylic acids are alternate lipid substrates in parenteral nutrition (Greco AV et al., Clin Nutr 1995, 14, 143). Basically, they are water soluble, undergo β-oxidation, do not induce ketogenesis but rather promote gluconeogenesis. They could represent an immediately available form of energy. Thus, inorganic salts of sebacic (C10) and dodecanedioic (C12) acids were firstly proposed, but now, triglycerides containing these fatty acids are under investigation (Capristo E et al. Clin Chim Acta 1999, 289, 11). Treatment of rats with derivatives of C16 dioic acid have shown that this compound markedly improved lipid metabolism (Russell JC et al., Arterioscl Thromb 1991, 11, 602) and inhibited the development of advanced cardiovascular disease (Russell JC et al., Arteriocl Thromb Vasc Biol 1995, 15, 918).
Bempedoic acid, a tetramethylated and hydroxylated dicarboxylic acid (a 15 carbon chain) is a drug under development (Esperion Therapeutics) for the treatment of hypercholesterolemia (Ray KK et al., N Engl J Med 2019, 380, 1022). This new drug inhibits ATP citrate lyase (after its activation to the thioester with coenzyme A) which is involved in the liver’s biosynthesis of cholesterol upstream of HMG-CoA reductase, the enzyme that is blocked by statins (Brandts J et al., Drugs 2020, 29, 763).
It has been shown that bempedoic acid provided additional LDL-C lowering in patients who did not achieve an adequate response to lipid-lowering therapy when compared with placebo (Goldberg AC et al., ). In 2020, the U.S. Food and Drug Administration considered it to be a first-in-class medication. The same year, the European Committee for Medicinal Products for Human Use has adopted a positive opinion, recommending the granting of a marketing authorisation for bempedoic acid under the name Nilemdo. That drug is used for the treatment of adults with primary hypercholesterolaemia or mixed dyslipidaemia when the recommended level of LDL-c may not be reached with statins. Reports have been published about the usefulness of that compound in the prevention of cardiovascular diseases (Warden BA et al., J Clin Lipidol 2022, 16, 94). In patients with statin intolerance as well as those without, bempedoic acid was shown to be a safe and efficacious lipid-lowering agent (Li Y et al., PLoS One 2024, 19, e0297854). In a randomized trial of 4206 statin-intolerant patients enrolled with high cardiovascular risk but without a prior cardiovascular event, bempedoic acid treatment, 180 mg daily, was associated with a significant reduction in major cardiovascular events (Nissen SE et al., JAMA 2023, 330, 13).
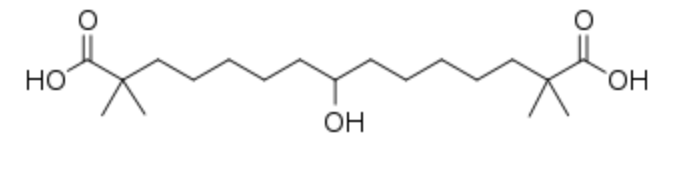 Bempedoic acid
Bempedoic acid
It must be recalled that the determination of the dicarboxylic acids generated by permanganate-periodate oxidation of monoenoic fatty acids was useful to study the position of the double bond in the carbon chain (Longmuir KJ et al., Anal Biochem 1987, 167, 213).
In 1900, Hildebrandt, who was studying the pharmacological effects of terpenes, reported the appearance of dicarboxylic acid derivatives of terpenes in the urine of rabbits treated with citral. Later, Kühn discovered that this substance was a dicarboxylic acid with a carboxy group at the ω-end of geranoic acid and is known as Hildebrandt acid. The presence of that compound in rabbit urine indicates the presence of an enzyme system in animal cells that oxidizes acyclic monoterpenoid alcohols to carboxylic acids (Shidoji Y, J Lipid Res 2023, 64, 100396
).
 Hildebrandt acid
Hildebrandt acid
Long-chain dicarboxylic acids containing vicinal dimethyl branching near the centre of the carbon chain have been discovered in the genus Butyrivibrio, bacteria which participate in the digestion of cellulose in the rumen (Klein RA et al., Biochem J 1979, 183, 691). These fatty acids, named diabolic acids, have a chain length depending on the fatty acid used in the culture medium. The most abundant diabolic acid in Butyrivibrio had a 32-carbon chain length.
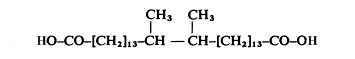
Diabolic acid (15,16-dimethyltriacontanedioic acid)
These diacids were also detected in he core lipids of the genus Thermotoga of the order Thermotogales, bacteria living in solfatara springs, deep-sea marine hydrothermal systems and high-temperature marine and continental oil fields (Huber R et al., Arch Microbiol 1986, 144, 324). It was shown that about 10% of their lipid fraction were symmetrical C30 to C34 diabolic acids. The C30 (13,14-dimethyloctacosanedioic acid) and C32 (15,16-dimethyltriacontanedioic acid) diabolic acids have been described in Thermotoga maritima (Caballeira NM et al., J Bacteriol 1997, 179, 2766).
Some parent C29 to C32 diacids but with methyl groups on the carbons C-13 and C-16 have been isolated and characterized from the lipids of thermophilic anaerobic eubacterium Themanaerobacter ethanolicus (Jung S et al., J Lipid Res 1994, 35, 1057). The most abundant diacid was the C30 α,ω-13,16-dimethyloctacosanedioic acid.
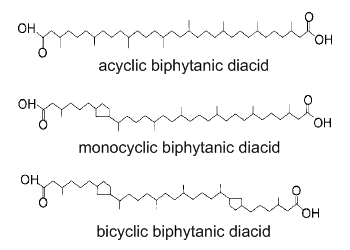
Biphytanic diacids are present in geological sediments and are considered as tracers of past anaerobic oxidation of methane (Birgel D et al., Org Geochem 2008, 39, 152). Several forms without or with one or two pentacyclic rings have been detected in Cenozoic seep limestones. These lipids may be unrecognized metabolites from Archaea.
Crocetin is the core compound of crocins (crocetin glycosides) which are the main red pigments of the stigmas of saffron (Crocus sativus) and the fruits of gardenia (Gardenia jasminoides). Crocetin is a 20-carbon chain dicarboxylic acid which is a diterpenenoid and can be considered as a carotenoid. It was the first plant carotenoid to be recognized as early as 1818 while the history of saffron cultivation reaches back more than 3,000 years.
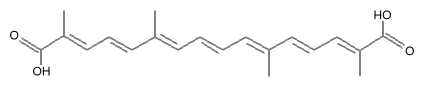
Crocetin
The major active ingredient of saffron is the yellow pigment crocin 2 (three other derivatives with different glycosylations are known) containing a gentiobiose (disaccharide) group at each end of the molecule.
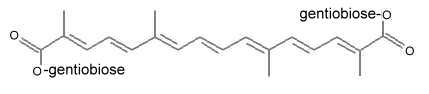
Crocin
A simple and speccific HPLC-UV method has been developed to quantify the five major biologically active ingredients of saffron, namely the four crocins and crocetin (Li N et al., J Chromatogr A 1999, 849, 349).
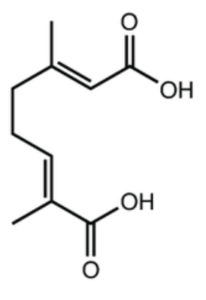
Several dicarboxylic acids having an alkyl side chain and an itaconate core have been isolated from lichens and fungi, itaconic acid (methylenesuccinic acid) being a metabolite produced by filamentous fungi.
Among these compounds, several analogues, called chaetomellic acids with different chain lengths and degrees of unsaturation have been isolated from various species of the lichen Chaetomella (two of them are shown below).
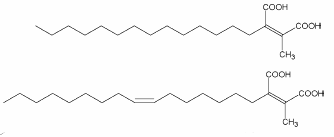
These molecules were shown to be valuable as basis for the development of anticancer drugs due to their strong farnesyltransferase inhibitory effects (Singh SB et al., Bioorg Med Chem 2000, 8, 571).
In 1999, a series of new fungal alkyl- and alkenyl-itaconates, ceriporic acids, were found in cultures of a selective lignin-degrading fungus (white rot fungus), Ceriporiopsis subvermispora (Enoki M et al., Chem Lett 2000, 54-55, Amirta R et al., Chem Phys Lipids 2003, 126, 121). Two of them are shown below.
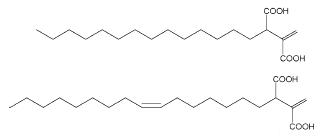
It was determined that these ceriporic acids suppressed iron redox reactions to attenuate •OH production by the Fenton reaction in the presence of iron reductants such as hydroquinone and cysteine (Enoki M et al., Chem Phys Lipids 2002, 120, 9). It was proposed that the suppression of the cellulolytic active oxygen species, •OH, by this metabolite contributes to the selective lignin-degradation with a minimum loss of cellulose. The absolute configuration of ceriporic acids, their stereoselective biosynthetic pathway and the diversity of their metabolites have been largely discussed (Nishimura H et al., Chem Phys Lipids 2009, 159, 77).
Dicarboxylic acids derive classically from medium and long-chain monocarboxylic fatty acids by an oxidation mechanism named w-oxidation which is followed in the cell by a b-oxidation. This w-oxidation is of minor significance in the normal cell physiology but the production of dicarboxylic acids may be increased by other mechanisms in particular situations of reaction-induced lipid peroxidation. It was shown that dicarboxylic acids may be the end products of oxidative attack on both free and esterified unsaturated fatty acids. The species formed are thus a distinctive feature of the positions of the double bonds in the intact fatty acid molecule. A novel cytotoxic phospholipid, 2-azelaoyl phospholipid, was thus described when phosphatidylcholines are peroxidized in vitro (Itabe H et al., Biochim Biophys Acta 1988, 962, 8).
The exact mechanism of dicarboxylic acid acid formation is not completely known but several experiments suggest that a single unsaturated fatty acid gives two types of dicarboxylic acids: a primary one related to the double bond nearest to the acid function and a secondary two carbon atoms lower homologue.
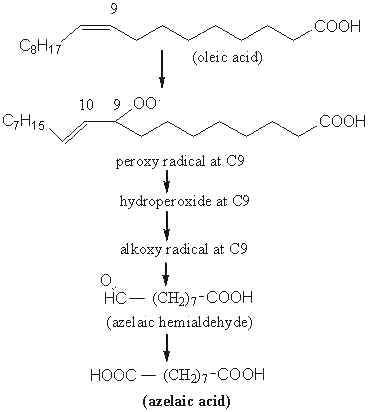
To illustrate the mechanism, the next figures represent the main steps of the oxidative attack of oleic acid (18:1 n-9) which will form azelaidic and pimelic acids.
First, oleic acid generates a peroxy radical at the carbon 9 and then may undergo a direct degradation to azelaic acid via the successive formation of peroxy, hydroperoxy and alkoxy radicals also at C9. The scission of the carbon chain at C9 will give an aldehyde-acid (azelaic hemialdehyde) and then a diacid (azelaidic acid).
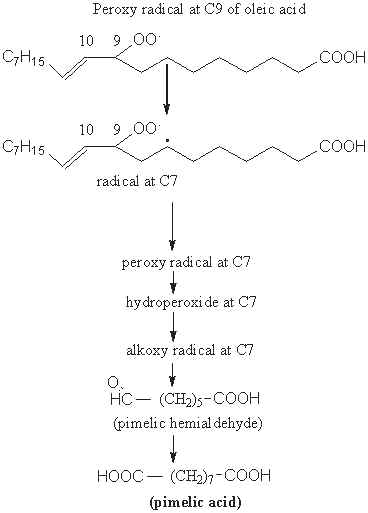
The second possibility is the formation of a peroxy radical at C9 followed by an intramolecular hydrogen transfer resulting in the formation of a radical at C7. A new sequence of reactions forms pimelic hemialdehyde and then the diacid, pimelic acid.
It was demonstrated that all free or esterified unsaturated fatty acids (mono- or polyunsaturated) under chemical, physical or biological oxidation generate dicarboxylic acids, their chain length depending on the position of the first double bond (Passi S et al., Biochim Biophys Acta 1993, 1168, 190).
Thus, linoleic acid (18:2n-6) may generate C9 and C7 diacids
α-linolenic acid (18:3n-3) may generate the same diacids
γ-linolenic acid (18:3n-6) may generate C6 and C4 diacids
arachidonic acid (20:4n-6) may generate C5 diacid (C3 was not detected)
EPA (20:5n-3) may generate the same diacid
DHA (22:6n-3) may generate C4 diacid.
It was shown that dicarboxylic acids may be use as markers of fatty acid peroxidation. Thus, in diabetes, where an imbalance between the generation of free radicals and antioxidant defense systems increases oxidative stress, the dicarboxylic acid excretion is increased in urine. It was shown that the urinary excretion increased by 5.5 and 7.5 times for adipic acid (C6) and suberic acid (C8), respectively (Inouye et al., Atherosclerosis 2000, 148, 197).
DISPERSIVE LIQUID-LIQUID MICROEXTRACTION
Lire la suiteDevenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire