
OILS USED IN DIETETICS, COSMETICS, AND LIPOCHEMISTRY
Some plant oils are used extensively in nutrition, cosmetics and lipochemistry:
Ahiflower (Buglossoides arvensis, Boraginacae). This species grows as both an annual and a biennial in habit. It is planted in early spring or in the autumn and takes from 100 – 150 days to reach maturity. Each flower can produce four seeds which are held tightly in the plant until harvest. This plant is pretty much endemic in the whole of the Northern Hemisphere and it has been seen as a weed species associated with the cultivation of wheat

Ahiflower (from naturescrops.com)
Seeds contain approximately 20% oil which offres the highest levels of omega-3 essential fatty acids among commercially available dietary plant oils. Approximately about 20% of the fatty acid content is stearidonic acid, 46% α-linolenic acid ALA) and 6% γ-linolenic acid, in addition to other beneficial fatty acids (7% oleic acid). Few seed oils containing stearidonic acid (SDA) have been produced for human consumption. Ribes nigrum seed oil (blackcurrant oil; 2–4 %) and Echium plantagineum seed oil (echium oil; 12–14 %) are produced commercially, and rapeseed and soyabeans have been genetically modified to produce seeds enriched in stearidonic acid. Thus, ahiflower combines the health support benefits of taking evening primrose, flaxseed, and probably fish oils, from a single plant. Ahiflower oil has been formulated into soft-gels, salad dressing, protein bars, plant-based milks and powders (www.ahiflower.com). Ahiflower oil produced exclusively by Nature’s Crops International (NCI) received certification from NSF International under its NSF Non-GMO True North protocol in April 2016. The US Food & Drug Administration (FDA) awarded the oil no-objection GRAS (generally recognised as safe) status in January 2015.
Consumption of this oil was shown to be safe and able to increases tissue long-chain n-3 fatty acid content more than flax seed oil, with efficiency for EPA but not for DHA (Lefort N et al., J Nutr Sci 2016, 5, e2). While the conversion of ALA into EPA is very low (about 3%), the conversion of SDA to EPA is higher (30-35%).According to the Ahiflower Division of TCI (Technology Crops International – part of Nature’s Crops International), a dose of approximately 2.3-3g/day of Ahiflower oil provides the recommended EPA minimum daily equivalent of 200-250mg.
Experiments on mice have shown that the distinct ahiflower oil composition could support tissue DHA needs at a similar rate to dietary DHA, making it a unique plant-based dietary option for maintaining DHA turnover comparably to dietary DHA (Metherel AH et al., Biochem Biophys Acta, Mol Cell Biol Lipids 2024, 1869, 159422). But could ahiflower oil eventually replace fish oil as a source of long-chain omega-3 fatty acids in human body as it is known inefficient in converting SDA to DHA, this is yet unresolved. It has been shown in 2024 that the consumption of ahiflower oil significantly improves the circulating levels of EPA and EPA-derived oxylipins in humans. In cultivated hepatocytes, EPA and EPA-derived oxylipins are more effectively increased by SDA than ALA (Seidel U et al., April 2024).
Avocado pear (Persea americana, Lauracae). The plant originated from America. It is mainly grown in USA, Mexico and Asia. Oil content of the fruit mesocarp: 8-22% (according to the variety and location). The world production of avocado oil is about 1,500 t. The oil is used in pharmacy and cosmetics, it is known to contain a high proportion of unsaponifiable substances (1-3 %) with a high percentage of sterols (20-70 % of the total), hydrocarbons (5-20 %) and triterpen alcohols (6-40 %).
The oil is mainly composed of oleic acid (67-72 %), palmitic acid (13-17 %), and linoleic acid (10-12 %). The main triglyceride species are OOP (24 %), OOO (32 %), LOO (10 %) and LOP (10 %) (Jakab A et al., J Chromatogr A 2002, 976, 255).
A chemical characterization of the oils from the pulp and seeds of avocado fruits may be found in the work by Bora PS et al. (Grasas y Aceites 2001, 52, 171). Data on glycolipids and phospholipids molecular species in the pulp and the almond of avocado have been reported (Pacetti D et al., J Chromatogr A 2007, 1150, 241).
Black-currant (Ribes nigrum, Saxifragacae). Frequently found in Europe and Asia. The oil, rich in γ-linolenic acid, is used in oral and parenteral nutrition, cosmetics and pharmacy (Patents by Nestlé SA). The composition of regioisomers of triacylglycerols of two currant oils was determined (Kallio H et al., Eur J Lipid Sci Technol 2005, 107, 101). It was shown that the preferential order of fatty acid to be in the sn-2 position was typically 18:1>18:2>18:4>18:3. If only 2.4% of the species contained three α– or γ-linolenic acids, about 18% contained two of these molecules. A typical fatty acid composition is : linoleic 38%, oleic 17.3%, linoleic 15.8%, γ-linolenic 14%, and palmitic 9% acids. The main triglyceride species are OLL 6.7%, LLL 6.3%, LLLn 6.2%, OLLn 5.1%, LLP 5%, gLnLP 5%, OLgLn 4.8%, and LLgLn 4% (Lisa M et al., J Chromatogr A 2008, 1198-1199, 11).

Black-currant
Borage (Borago officinalis, Boraginacae). Oil is rich in γ-linolenic acid (18-25%). Cultivated in west mediterranean regions. Oil content: 13-33%. A typical fatty acid composition is : linoleic (35.4%), oleic (22.9%), γ-linolenic (18.4%), and palmitic (11%) acids.
The oil is used in pharmacy and cosmetic.

Borage
Cacao (Theobroma cacao, Sterculiacae). The plant originated from Amazonia but is cultivated in Africa and Brazil.
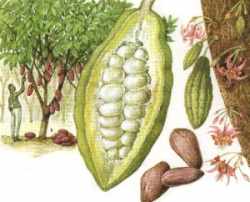
Fat content of the roasted kernels: 50-60%. The roasted bean is ground to give cacao mass from which much of the cacao butter is extracted by pressing. Its melting point is about 33°C and is composed of 24-29% palmitic acid, 34-36% stearic acid,30-40% oleic acid and 2.4% linoleic acid. The main triglyceride species are SOP (36.4%), SOS (23.5%), and POP (16.6%) (Lisa M et al., J Chromatogr A 2008, 1198-1199, 115). Both for cocoa and chocolates differences in fatty acid profile are observed and are mainly explained as an effect of the geographical origin, and not by processing conditions. For cocoa, differences in fatty acid profile are found in C12:0, C14:0, C16:0, C16:1, C17:0, C17:1 and C18:0 (Torres-Moreno M., et al., Food Chem 2015, 166, 125).
Cacao butter is used in human nutrition (manufacture of chocolate), pharmacy (suppositories) and cosmetics. The world production of cacao beans was about 3 million tons in 1999.
The uncertainty in cocoa butter supply and the volatility of its price have forced producers to seek alternatives to replace cocoa butter. Thus many similar fats were used as confectionery fats such as lauric cocoa butter substitutes, non-lauric cocoa butter substitutes and cocoa butter equivalents. The last ones are closest to cocoa butter. The European Union requirements are : SOS > ou = to 65%, unsaturated FA at the position sn-2 > ou = to 85%, total unsaturated FA < or = to 45%. The use of specific lipases and various source oils enable a large-scale production of cocoa butter substitutes.
Castor bean (Ricinus communis, Euphorbiacae). Castor was used in Egypt as a lamp oil about 6000 years ago. Later, it was used as a cosmetic product and in medicine (laxative) in India and China. It is indigenous to eastern Africa, and most probably originated in Ethiopia.

Oil content: about 50%. The oil is composed of more than 94% triacylglycerols, the remaining being monoglycerides (3.4%), and phospholipids (0.6%). Castor oil is the only commercial source of ricinoleate. Its specific fatty acid composition is : 1% 18:0, 3% 18:1, 3% 18:2, about 90% hydroxylated 18:1 (ricinoleic acid) and 1% dihydroxylated 18:2. About 70% of castor oil is tri-ricinolein (tri-ricinoleoylglycerol) (Lin JT et al., J Liq Chromatogr Relat Technol 2003, 26, 773). Moreover, 0.5% of the lipid content is composed of a tetra-acylglycerol containing four ricinoleic acid molecules (12-ricinoleoylricinoleoyl) di-ricinoleoylglycerol), 95% of the 12-ricinoleoylricinoleoyl chain being identified at the sn-2 position of the glycerol backbone (Lin JT et al., J Agric Food Chem 2006, 54, 3498).
Castor oil and many of its derivatives are stabilized by the hydroxyl group, which is beta to the double bond. This hydroxyl group protects the double bond by preventing the formation of peroxides. As a result, castor oil is approximately four times more stable than olive oil. The oil is more dense and viscous than most other vegetable oils and completely miscible with ethanol.
After suitable pretreatment, the oil is poorly used in medicine but largely used in lipochemistry (drying oils, paints, ink, soaps, lubricants, varnish), cosmetics (non-comedogens and emollients) and food industry. Chemical industry used castor oil after oxidation, alkoxylation, dehydration, hydrogenation and sulfonation.
In the early years of the 20th century, castor oil was found to be a good lubricant for rotary aircraft engines. It’s likely that the hydroxyl groups confer the properties that make it a much better lubricant than other vegetable oils. The castor-containing lubricants developed and sold by the Wakefield Oil Company was so successful that the name of the company was changed into Castrol which sells motor oils to this day.
The world production of castor beans was about 0.5 million tons in 2004-05, India producing about 70% of the total, the remaining being produced in China (18%), Brazil, Russia, Thailand and some African countries.
Evening primrose (Oenothera biennis, Onagracae). Oil well known for its high content in γ-linolenic acid. Numerous varieties are cultivated in America and in Europe. Oil content: 12-30%. The oil is used in pharmacy and cosmetics.
Kukui oil (Aleurites moluccana, Euphorbiacae). The tree which gives the nuts is the official symbol of the “Aha Kupuna, the Council of Elders of the Nation of Hawaii”. The seeds contain a high amount of fluid oil which has many uses, among them lighting (the tree is also named “candlenut tree”), in medicine (purgative, psoriasis, eczema and other dry skin problems), lipochemistry (varnish) and in cosmetics. The oil is said to contain 25.6% linolenic acid, 39.4% linoleic acid, 25% oleic acid, 9% palmitic acid. The main triglyceride species are OLLn (13.6%), LLLn (11%), OLL (10.2%), LLL (7.5%), LnLLn (6.6%) (Lisa M et al., J Chromatogr A 2008, 1198-1199, 115).
Pumpkin seed (Cucurbita pepo). The oil is used in cooking and dietetics. It is a culinary specialty in countries related to the ancient Austro-Hungarian Empire. It has an intense nutty taste and is rich in polyunsaturated fatty acids. Pumpkin seed oil serves as a salad dressing. Pumpkin seed oil is said to help promote urinary health and heart health. It’s full of vitamins and anti-inflammatory agencies, along with linoleic (36-63%) and oleic acid (17-39%). Its content in saturated fatty acids is low (16:0 12-18%, 18:0 5-8.5%, 20:0 0.3-1.1%). Pumpkin seed oil, along with palmetto oil, has shown promising results as an alternative therapy for benign prostate hypertrophy (Hong H et al., Nutr Res Pract 2009, 3, 323; Vahlensieck W et al., Urol Int 2015, 94, 286).
Wheat germ (Triticum vulgare, Graminae). The oil is used in dietetics and cosmetic. A typical fatty acid composition is linoleic (54.8%), oleic (17.8%), palmitic (16.5%) and linolenic (8%) acids. The main triglyceride species are LLP (19.8%), LLL (15.3%),OLL (13.5%), and LLLn (6.5%) (Lisa M et al., J Chromatogr A 2008, 1198-1199, 115).
Devenez membre et participez au développement de la Lipidomique au XXIème siècle.
S'inscrire